Hikers who found a human body at high altitude in the Italian Alps thought the man had died recently, but tests indicated he was shot with an arrow more than 5,300 years ago.
Which of the following would be the best reason for prolonged preservation of the body? .
The food that the person ate contained toxins that killed the bacteria that would have otherwise destroyed the body
The arrow wound caused blood to flow out of the body, which led the enzymes that would break down tissue to be cleared from the body
The body was frozen in the cold temperature of the Alps shortly after he died and remained frozen until it was found
The ultraviolet rays at such a high altitude caused all the body's molecules to be preserved.
Correct Answer : C
The best reason for the prolonged preservation of the body is that it was frozen in the cold temperature of the Alps shortly after he died and remained frozen until it was found.
Freezing can preserve a body by slowing down or stopping the decomposition process.
Choice A is not correct because the food that the person ate would not have contained toxins that killed the bacteria that would have otherwise destroyed the body.
Choice B is not correct because the arrow wound would not have caused blood to flow out of the body in a way that would have cleared enzymes that break down tissue from the body.
Choice D is not correct because ultraviolet rays at high altitude would not have caused all of the body’s molecules to be preserved.
TEAS 7 Exam Quiz Bank
HESI A2 Exam Quiz Bank
Find More Questions 📚
Teas 7 Questions: We got the latest updated TEAS 7 questions
100% Money Refund: 100% money back guarantee if you take our full
assessment pass with 80% and fail the actual exam.
Live Tutoring: Fully customized live tutoring lessons.
Guaranteed A Grade: All students who use our services pass with 90%
guarantee.
Related Questions
Correct Answer is B
Explanation
The atomic number of an element represents the number of protons in the nucleus of an atom of that element.
Since lithium has an atomic number of 3, it has 3 protons in its nucleus.
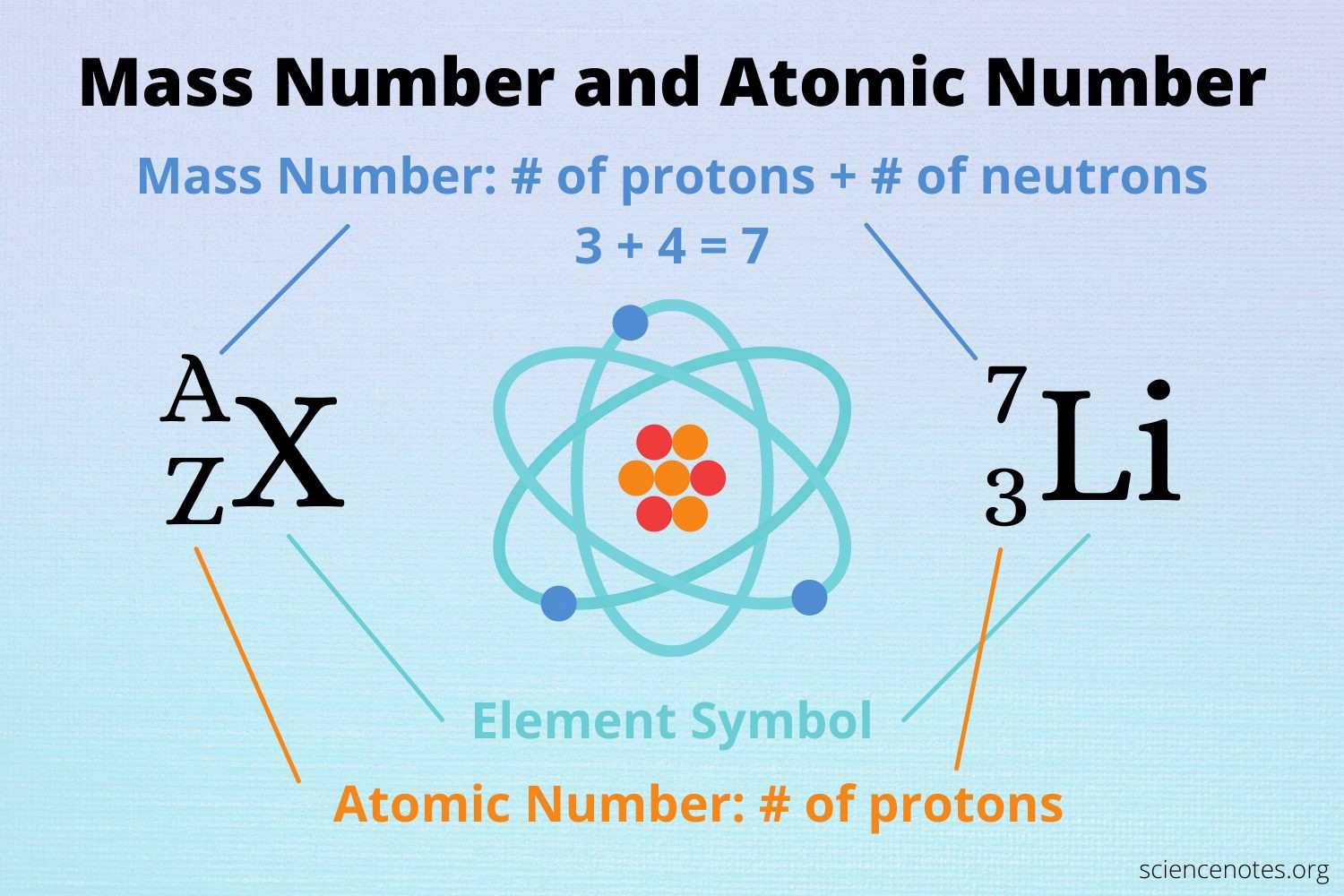
Choice A is not correct because 7 is the mass number of lithium, not the number of protons.
Choice C is not correct because 12 is not the atomic number or mass number of lithium.
Choice D is not correct because 4 is not the atomic number or mass number of lithium.
Correct Answer is C
Explanation
The pH scale is a logarithmic scale that measures the acidity or alkalinity of a solution.
A solution with a pH of 7 is neutral, while a solution with a pH less than 7 is acidic and a solution with a pH greater than 7 is alkaline.
Because the pH scale is logarithmic, each whole number change in pH represents a tenfold change in acidity or alkalinity.
Therefore, a substance with a pH of 3 is 10 times more acidic than a substance with a pH of 4.
Choice A.
A substance with a pH of 3 is two times more alkaline than a substance with a pH of 4 is not correct because it incorrectly states that the substance with a lower pH is more alkaline and also incorrectly states the magnitude of the difference in acidity or alkalinity.
Choice B.
A substance with a pH of 3 is two times more acidic than a substance with a pH of 4 is not correct because it correctly states that the substance with a lower pH is more acidic but incorrectly states the magnitude of the difference in acidity.
Choice D.
A substance with a pH of 3 is 10 times more alkaline than a substance with a pH of 4 is not correct because it incorrectly states that the substance with a lower pH is more alkaline.
Correct Answer is B
Explanation
Diffusion down a concentration gradient causes most of the carbon dioxide from the blood to move into the alveoli.
The alveoli are tiny air sacs in the lungs where gas exchange occurs.
Carbon dioxide is a waste product of cellular respiration and is carried by the blood to the lungs to be exhaled.
In the lungs, carbon dioxide diffuses from the blood (where its concentration is high) into the alveoli (where its concentration is lower) down its concentration gradient.
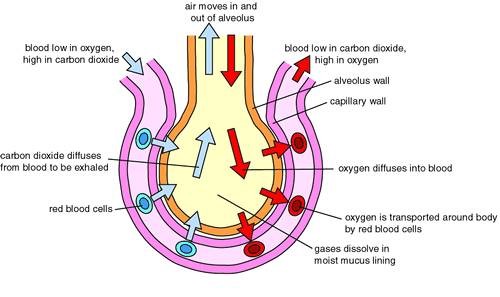
Choice A is incorrect because carbon dioxide is not converted to carbon monoxide in the body.
Choice C is incorrect because passive transport using carrier proteins is not the primary mechanism by which carbon dioxide moves from the blood into the alveoli.
Choice D is incorrect because active transport using energy is not involved in the movement of carbon dioxide from the blood into the alveoli.
Correct Answer is B
Explanation
Electrophoresis is the most useful laboratory method for separating genomic DNA fragments by size.
Electrophoresis is a technique that uses an electric field to separate charged molecules, such as DNA fragments, based on their size and charge.
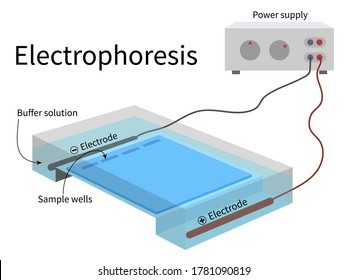
Choice A is not correct because titration is a laboratory method used to determine the concentration of a solution.
Choice C is not correct because filtration is a laboratory method used to separate solids from liquids.
Choice D is not correct because spectrophotometry is a laboratory method used to measure the absorbance of light by a solution.
Correct Answer is D
Explanation
Triple covalent bonds.
Nitrogen gas (N2) is an extremely stable molecule because it consists of two nitrogen atoms bonded together by a triple covalent bond.
A covalent bond is a type of chemical bond where atoms share electrons to form a molecule.
In a triple covalent bond, three pairs of electrons are shared between the two atoms, resulting in a very strong bond that makes the molecule extremely stable.
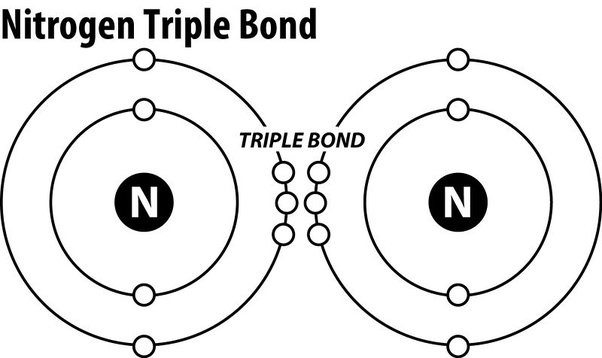
Choice A.
Ionic bonds is not correct because ionic bonds involve the transfer of electrons from one atom to another to form ions, which are then attracted to each other due to their opposite charges.
Nitrogen gas does not contain ions and is not held together by ionic bonds.
Choice B.
Hydrogen bonds is not correct because hydrogen bonds are weak electrostatic attractions between molecules that contain hydrogen atoms bonded to highly electronegative atoms such as oxygen or nitrogen.
Nitrogen gas does not contain hydrogen atoms and is not held together by hydrogen bonds.
Choice C.
Resonance bonds is not correct because resonance refers to the delocalization of electrons in a molecule where multiple Lewis structures can be drawn to represent the molecule.
Nitrogen gas has a single Lewis structure and does not exhibit resonance.
Correct Answer is A
Explanation
The approximate threshold value for mammalian neurons is -55 mV.
The threshold potential is the critical level to which a membrane potential must be depolarized to initiate an action potential.
Most often, the threshold potential is a membrane potential value between –50 and –55 mV
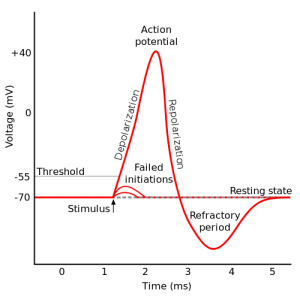
The membrane potential of a neuron is determined by the distribution of ions across the cell membrane.
At rest, the inside of a neuron is more negative than the outside due to the presence of negatively charged proteins and other molecules.
The movement of ions across the cell membrane can change the membrane potential.
For example, when sodium ions enter the cell, they make the inside of the cell more positive (less negative), causing depolarization.
Choice B is incorrect because -80 mV is below the typical threshold value for mammalian neurons.
Choice C is incorrect because +35 mV is above the typical threshold value for mammalian neurons.
Choice D is incorrect because 0 mV is above the typical threshold value for mammalian neurons.
Correct Answer is C
Explanation
Amino acids have a unique structure consisting of an amino group (-NH3⁺) and a carboxyl group (-COO⁻) attached to a central carbon (called the α-carbon). At physiological pH (around 7.4), these functional groups often exist in their ionized forms:
- The amino group (-NH3⁺) is positively charged, acting as a proton acceptor (a base).
- The carboxyl group (-COO⁻) is negatively charged, acting as a proton donor (an acid).
This results in a zwitterion — a molecule with both a positive and a negative charge. Because amino acids can accept or donate protons depending on the pH of their environment, they have buffering capacity. This means they can resist changes in pH by stabilizing the concentration of hydrogen ions (H⁺).
Why Other Options Are Incorrect:
- A. Monosaccharides: These are simple sugars without ionizable functional groups, so they cannot act as buffers.
- B. Ribonucleotides and D. Deoxyribonucleotides: While nucleotides have phosphate groups that can donate protons, they lack the dual positive and negative functional groups necessary for the strong buffering effect seen in amino acids.
Therefore, amino acids are the correct choice because their zwitterionic nature provides them with excellent buffering capacity.
Correct Answer is D
Explanation
Viruses.
Viruses lack essential machinery needed to reproduce by themselves.
In fact, viruses can only reproduce after infecting a living cell - a process called viral replication.
Once inside a living cell, viruses re-program the cell’s machinery to produce viral proteins and genetic material to make new copies of themselves.
Choice A, Bacteria, is not the correct answer because bacteria have their own metabolic pathways and can reproduce outside of a host cell.
Choice B, Protozoa, is also not the correct answer because protozoa are singlecelled eukaryotes that have their own metabolic pathways and can reproduce outside of a host cell.
Choice C, Helminths, is not the correct answer because helminths are multicellular parasitic worms that have their own metabolic pathways and can reproduce outside of a host cell.
Correct Answer is D
Explanation
Testosterone is classified as an androgen hormone.
Androgens are a type of sex hormone that primarily regulates the development and maintenance of male characteristics, such as body hair growth, muscle mass, and deepening of the voice.
Testosterone is produced primarily in the testes in males and in smaller amounts in the ovaries and adrenal glands in females.
Option A, estrogen, is a female hormone that regulates the development of female sexual characteristics, such as breast growth and menstruation.
While estrogen and testosterone are both steroid hormones and can be converted to one another in the body, testosterone is not categorized as estrogen.
Option B, progestin, is a synthetic form of the hormone progesterone.
Progesterone is a female hormone that plays a role in the menstrual cycle and pregnancy.
Testosterone and progestin are not related, and testosterone is not categorized as progestin.
Option C, aldosterone, is a mineralocorticoid hormone that regulates salt and water balance in the body.
It is produced in the adrenal gland and plays a role in regulating blood pressure.
Testosterone and aldosterone are not related, and testosterone is not categorized as aldosterone.
Correct Answer is B
Explanation
The atomic number of an atom is equal to the number of protons in its nucleus.
In this case, the atom has 12 protons, so its atomic number is 12.
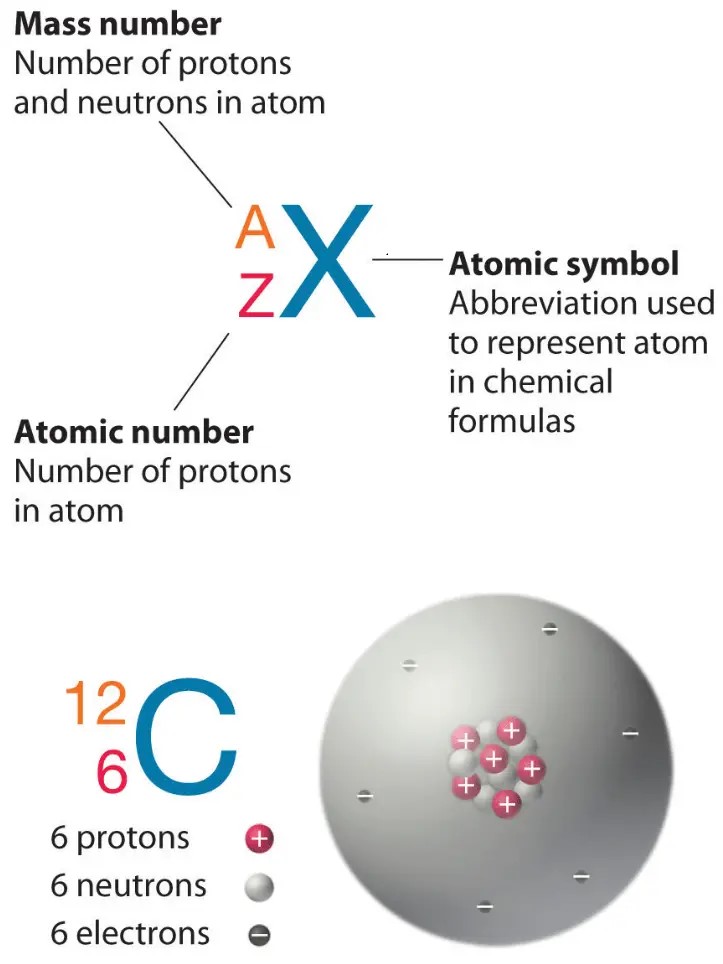
Choice A, 24, is not the correct answer because it represents the sum of the number of protons and neutrons in the atom’s nucleus, which is known as the mass number.
Choice C, 1, is not the correct answer because it does not represent the number of protons in the atom’s nucleus.
Choice D, 144, is not the correct answer because it represents the square of the mass number and does not represent any property of the atom.
This question was extracted from the actual TEAS Exam. Ace your TEAS exam with the actual TEAS 7 questions, Start your journey with us today
Visit Naxlex, the Most Trusted TEAS TEST Platform With Guaranteed Pass of 90%.
Money back guarantee if you use our service and fail the actual exam. Option of personalised live tutor on your area of weakness.
