In the following data table of an experiment carried out at 4°C (39.2 F) over 4 hours
|
Solution in bag |
Solution outside bag |
Bag mass change (g): |
|
water |
Water |
-0.2 |
|
20% sucrose |
Water |
+2.4 |
|
, 40% sucrose |
Water |
+4.3 |
|
, 60% sucrose |
water |
+5.8 |
Which of the following options represents the dependent variable?
Duration
Temperature
Bag mass change
Solution used outside
Correct Answer : C
Bag mass change is the dependent variable in this experiment.
In an experiment, the dependent variable is the variable that is being measured and is expected to change in response to changes in the independent variable(s).
In this case, the bag mass change is being measured and is expected to change in response to changes in the independent variable (sucrose concentration).
Choice A is incorrect because duration is not a variable in this experiment.
Choice B is incorrect because temperature is not a variable in this experiment.
Choice D is incorrect because sucrose concentration is an independent variable, not a dependent variable.
An independent variable is a variable that is manipulated by the experimenter to see how it affects the dependent
TEAS 7 Exam Quiz Bank
HESI A2 Exam Quiz Bank
Find More Questions 📚
Teas 7 Questions: We got the latest updated TEAS 7 questions
100% Money Refund: 100% money back guarantee if you take our full
assessment pass with 80% and fail the actual exam.
Live Tutoring: Fully customized live tutoring lessons.
Guaranteed A Grade: All students who use our services pass with 90%
guarantee.
Related Questions
Correct Answer is A
Explanation
The glomerulus is the main filtering unit of the kidney.
It is formed by a network of small blood vessels (capillaries) enclosed within a sac called the Bowman’s capsule.
The blood supply to the glomerulus is provided via the afferent arteriole.
The blood then flows through the capillary network, where it gets filtered, and then leaves the glomerulus via the efferent arteriole.
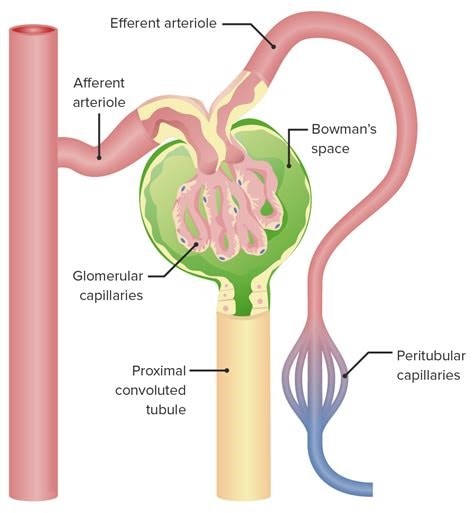
Choice B.
Proximal tubule is not correct because it is where the ultrafiltrate collected in the Bowman’s space drains directly into.
Choice C.
Distal tubule is not correct because it is not mentioned in relation to blood exiting the glomerulus.
Choice D.
Afferent arteriole is not correct because it provides blood supply to the glomerulus.
Correct Answer is C
Explanation
Control.
A control group is a group in an experiment that does not receive the treatment or manipulation being tested and is used as a benchmark to measure how the other tested subjects do.
The control group is used to minimize the effects of all variables except the independent variable.
This allows researchers to determine if changes in the dependent variable are due to the manipulation of the independent variable or if they are due to some other factor.
Choice A.
Responding is not the correct answer because it refers to the dependent variable, which is the variable that is being measured in an experiment.
Choice B.
Manipulated is not the correct answer because it refers to the independent variable, which is the variable that is being manipulated in an experiment.
Choice D.
Variable is not the correct answer because it refers to any factor that can change in an experiment and can include both independent and dependent variables.
Correct Answer is C
Explanation
Carbonic acid.
In the human body, maintaining the pH of the blood within a narrow range is critical for proper physiological functioning.
One of the buffering systems that helps to regulate blood pH involves the conversion of carbon dioxide (CO2) and water (H2O) into carbonic acid (H2CO3), which then dissociates into hydrogen ions (H+) and bicarbonate ions (HCO3-).
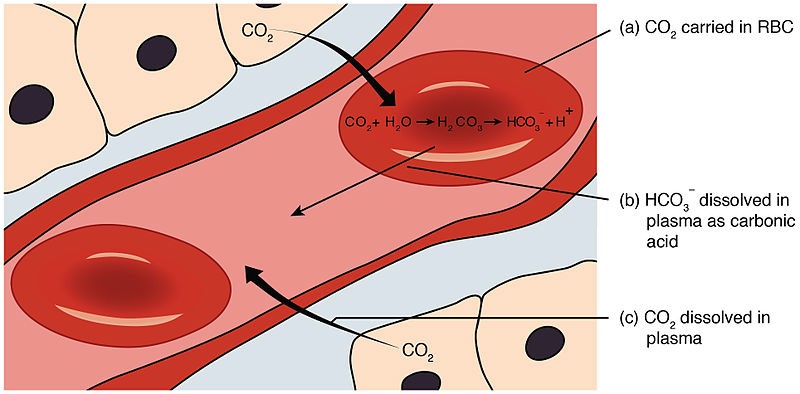
Carbonic acid (H2CO3) is responsible for donating H+ ions to act as a buffer when blood pH rises.
When blood pH rises (becomes more alkaline), carbonic acid dissociates, and the H+ ions combine with bicarbonate ions to form more carbonic acid.
This helps to remove excess H+ ions from the blood and prevent the pH from rising too much.
Option A, carbon dioxide, is involved in the buffering system through its conversion to carbonic acid.
However, it does not directly donate H+ ions to act as a buffer when blood pH rises.
Option B, carbon monoxide, is a toxic gas that binds to hemoglobin in red blood cells, preventing them from carrying oxygen.
It is not involved in the buffering system and does not donate H+ ions.
Option D, oxygen, is carried by hemoglobin in red blood cells and is essential for respiration.
It is not involved in the buffering system and does not donate H+ ions.
Correct Answer is D
Explanation
Triple covalent bonds.
Nitrogen gas (N2) is an extremely stable molecule because it consists of two nitrogen atoms bonded together by a triple covalent bond.
A covalent bond is a type of chemical bond where atoms share electrons to form a molecule.
In a triple covalent bond, three pairs of electrons are shared between the two atoms, resulting in a very strong bond that makes the molecule extremely stable.
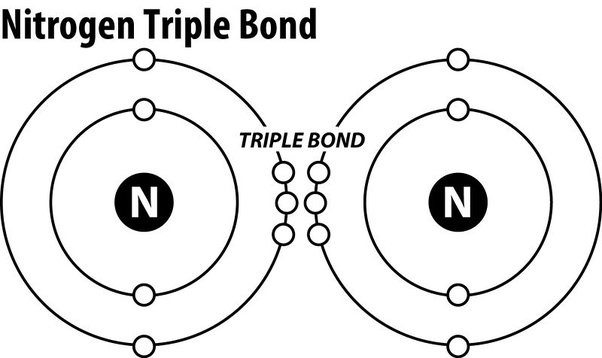
Choice A.
Ionic bonds is not correct because ionic bonds involve the transfer of electrons from one atom to another to form ions, which are then attracted to each other due to their opposite charges.
Nitrogen gas does not contain ions and is not held together by ionic bonds.
Choice B.
Hydrogen bonds is not correct because hydrogen bonds are weak electrostatic attractions between molecules that contain hydrogen atoms bonded to highly electronegative atoms such as oxygen or nitrogen.
Nitrogen gas does not contain hydrogen atoms and is not held together by hydrogen bonds.
Choice C.
Resonance bonds is not correct because resonance refers to the delocalization of electrons in a molecule where multiple Lewis structures can be drawn to represent the molecule.
Nitrogen gas has a single Lewis structure and does not exhibit resonance.
Correct Answer is D
Explanation
The polarity of water molecules explains its solvent abilities for certain substances.
Water is a polar molecule because it has a partial positive charge on one end and a partial negative charge on the other end due to the unequal sharing of electrons between the oxygen and hydrogen atoms.
This polarity allows water to dissolve other polar substances and ionic compounds.
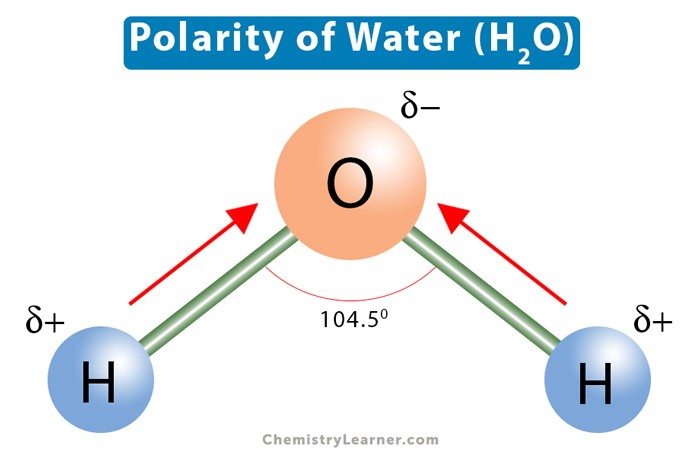
Choice A.
Kinetic energy of liquid water molecules is not the correct answer because kinetic energy refers to the energy of motion and does not directly explain water’s solvent abilities.
Choice B.
High specific heat is not the correct answer because specific heat refers to the amount of heat required to raise the temperature of a substance and does not directly explain water’s solvent abilities.
Choice C.
High surface tension is not the correct answer because surface tension refers to the cohesive forces between liquid molecules and does not directly explain water’s solvent abilities.
Correct Answer is C
Explanation
Ovulation is the process in which an ovarian follicle matures and releases a reproductive egg.
During ovulation, a mature egg is released from the female ovary, enabling it to be fertilized by male sperm cells 1.
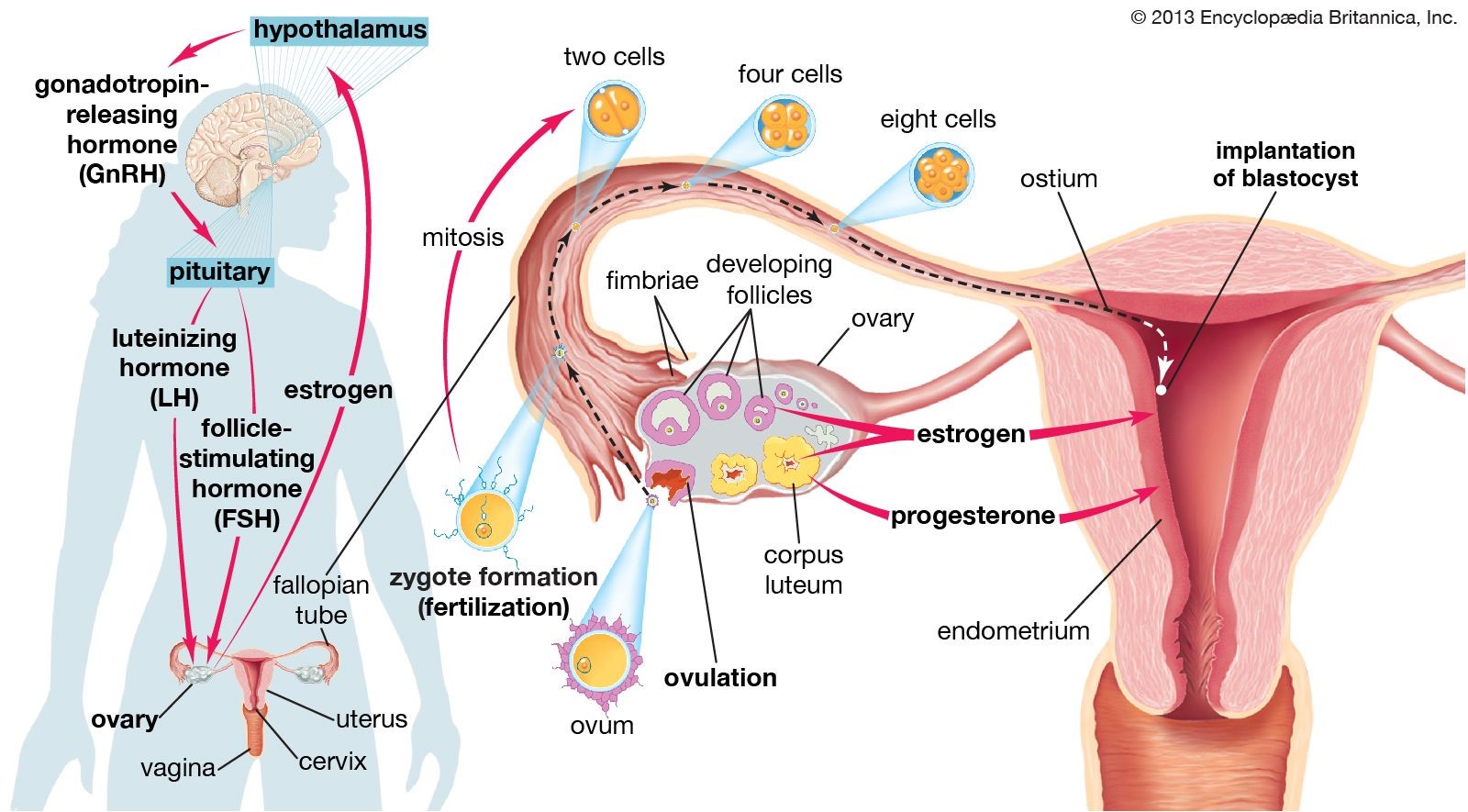
Choice A is incorrect because menstruation is the process of shedding the uterine lining, which occurs when an egg is not fertilized.
Choice B is incorrect because fertilization is the process of a sperm cell joining with an egg cell to form a zygote.
Choice D is incorrect because oogenesis is the process of forming female gametes (eggs) in the ovaries.
Correct Answer is B
Explanation
Electrophoresis is the most useful laboratory method for separating genomic DNA fragments by size.
Electrophoresis is a technique that uses an electric field to separate charged molecules, such as DNA fragments, based on their size and charge.
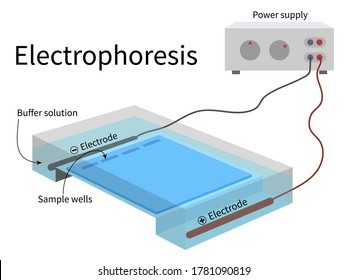
Choice A is not correct because titration is a laboratory method used to determine the concentration of a solution.
Choice C is not correct because filtration is a laboratory method used to separate solids from liquids.
Choice D is not correct because spectrophotometry is a laboratory method used to measure the absorbance of light by a solution.
Correct Answer is D
Explanation
A totipotent cellcan self-renew by dividing and develop into the three primary germ cell layers of the early embryo and into extra-embryonic tissues such as the placenta.
A fertilized egg is a totipotent stem cell and as such can develop into any specialized cell found in the organism.
Choice A is not correct because totipotent cells do not fight infectious diseases.
Choice B is not correct because totipotent cells do not aid in the maturation of sex cells.
Choice C is not correct because totipotent cells do not carry electrical impulses.
Correct Answer is C
Explanation
A tsunami is a catastrophic ocean wave that is usually caused by a submarine earthquake.
It can also be caused by an underwater or coastal landslide, the eruption of a volcano, or the impact of a meteor or comet in a body of water.
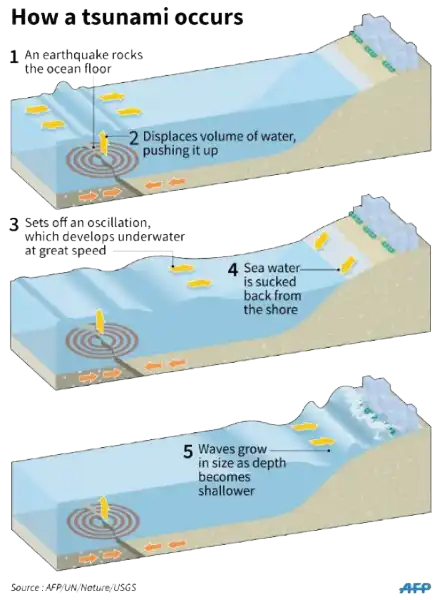
Choice A is not correct because sunspot activity does not cause tsunamis.
Choice B is not correct because lightning strikes do not cause tsunamis.
Choice D is not correct because flooding does not cause tsunamis.
Correct Answer is D
Explanation
The pulmonary veins are the vessels that carry oxygenated blood from the lungs to the left atrium of the heart.
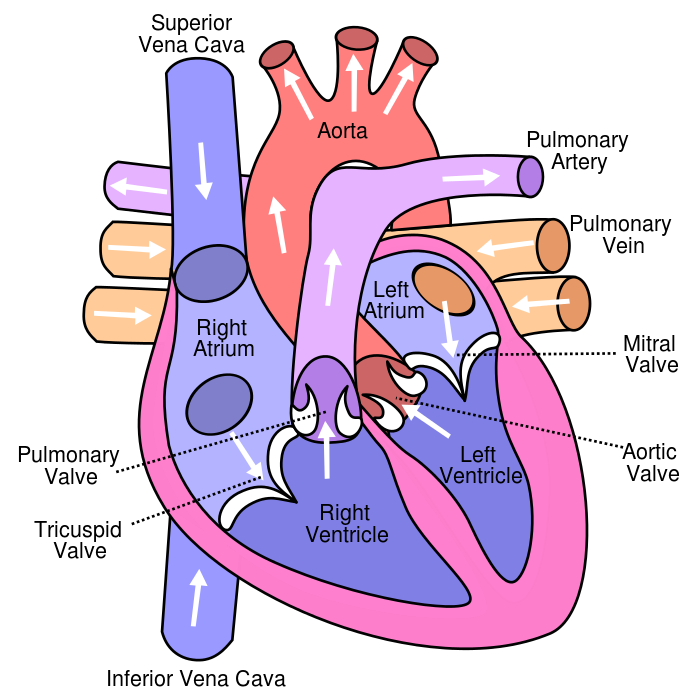
Choice A is not correct because the superior vena cava carries deoxygenated blood from the upper body to the right atrium of the heart.
Choice B is not correct because the inferior vena cava carries deoxygenated blood from the lower body to the right atrium of the heart.
Choice C is not correct because the pulmonary artery carries deoxygenated blood from the right ventricle of the heart to the lungs.
This question was extracted from the actual TEAS Exam. Ace your TEAS exam with the actual TEAS 7 questions, Start your journey with us today
Visit Naxlex, the Most Trusted TEAS TEST Platform With Guaranteed Pass of 90%.
Money back guarantee if you use our service and fail the actual exam. Option of personalised live tutor on your area of weakness.
