In the following single-replacement reaction, ______ replaces ______.
Cl2+2NaI→2NaCl+I2
sodium, iodine
chlorine, iodine
chlorine, sodium
sodium, chlorine
Correct Answer : B
In this reaction, chlorine (Cl2) is an element in the reaction that replaces iodine in the compound sodium iodide (NaI). This allows chlorine to form a compound with sodium (NaCl) and leaves iodine (I2) as an element.
Synthesis reactions involve two or more reactants (A and B) combining to form one product (AB). In the example provided, hydrogen (H2) and oxygen (O2) begin as separate elements. At the end of the reaction, the hydrogen and oxygen atoms are bonded in a molecule of water (H2O).
Decomposition reactions have only one reactant (AB) that breaks apart into two or more products (A and B). In the example above, hydrogen peroxide (H2O2) breaks apart into two smaller molecules: water (H2O) and oxygen (O2).
Single-replacement reactions involve two reactants, one compound (AB) and one element (C). In this type of reaction, one element replaces another to form a new compound (AC), leaving one element by itself (B). In the example, zinc replaces hydrogen in hydrochloric acid (HCl). As a result, zinc forms a compound with chlorine, zinc chloride (ZnCl2), and hydrogen (H2) is left by itself.
Double-replacement reactions involve two reactants, both of which are compounds made of two components (AB and CD). In the example, silver nitrate, composed of silver (Ag1+) and nitrate (NO31-) ions, reacts with sodium chloride, composed of sodium (Na1+) and chloride (Cl1-) ions. The nitrate and chloride ions switch places to produce two compounds that are different from those in the reactants.
Combustion reactions occur when fuels burn, and they involve specific reactants and products, as seen in the examples below. Some form of fuel that contains carbon and hydrogen is required. Examples of such fuels are methane, propane in a gas grill, butane in a lighter, and octane in gasoline. Notice that these fuels all react with oxygen, which is necessary for anything to burn. In all combustion reactions, carbon dioxide, water, and energy are produced. When something burns, energy is released, which can be felt as heat and seen as light.
TEAS 7 Exam Quiz Bank
HESI A2 Exam Quiz Bank
Find More Questions 📚
Teas 7 Questions: We got the latest updated TEAS 7 questions
100% Money Refund: 100% money back guarantee if you take our full
assessment pass with 80% and fail the actual exam.
Live Tutoring: Fully customized live tutoring lessons.
Guaranteed A Grade: All students who use our services pass with 90%
guarantee.
Related Questions
Correct Answer is D
Explanation
A person can be a universal blood donor or acceptor. A universal blood donor has type O blood, while a universal blood acceptor has type AB blood.
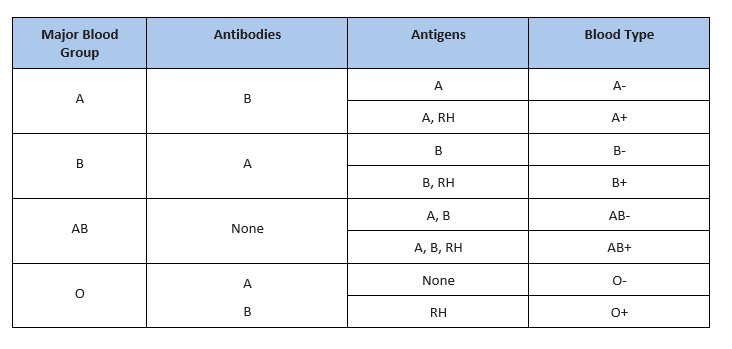
There are several different types or groups of blood, andthe major groups are A, B, AB, and O. Blood group is a way to classify blood according to inherited differences of red blood cellantigensfound on the surface of a red blood cell. The type ofantibodyin blood also identifies a particular blood group. Antibodies are proteins found in the plasma. They function as part of the body’s natural defense to recognize foreign substances and alert the immune system.

Depending on which antigen is inherited, parental offspring will have one of the four major blood groups. Collectively, the following major blood groups comprise the ABO system:
- Blood group A: Displays type A antigens on the surface of a red blood cell and contains B antibodies in the plasma.
- Blood group B: Displays type B antigens on the red blood cell’s surface and contains A antibodies in the plasma.
- Blood group O: Does not display A or B antigens on the surface of a red blood cell. Both A and B antibodies are in the plasma.
- Blood group AB: Displays type A and B antigens on the red blood cell’s surface, but neither A nor B antibodies are in the plasma
In addition to antigens, the Rh factor protein may exist on a red blood cell’s surface. Because this protein can be either present (+) or absent (-), it increases the number of major blood groups from four to eight: A+, A-, B+, B-, O+, O-, AB+, and AB-.
Correct Answer is D
Explanation
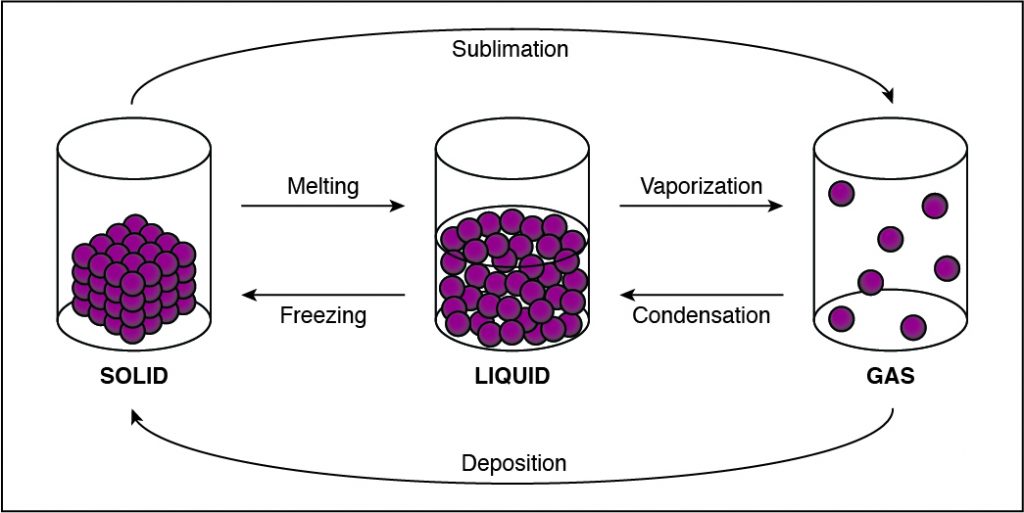
If the cohesion between particles decreases, then the particles must be undergoing a phase change that allows particles to move farther apart. This happens when a substance vaporizes and turns from liquid to gas. Any phase change that moves to the right in the diagram above requires energy to be added to the system because the substance has more energy at the end of the phase change. The phase changes aremelting,vaporization (boiling), andsublimation. When energy is added, particles move faster and can break away from each other more easily as they move to a state of matter with a higher amount of energy. This is most commonly done by heating the substance.
Correct Answer is A
Explanation
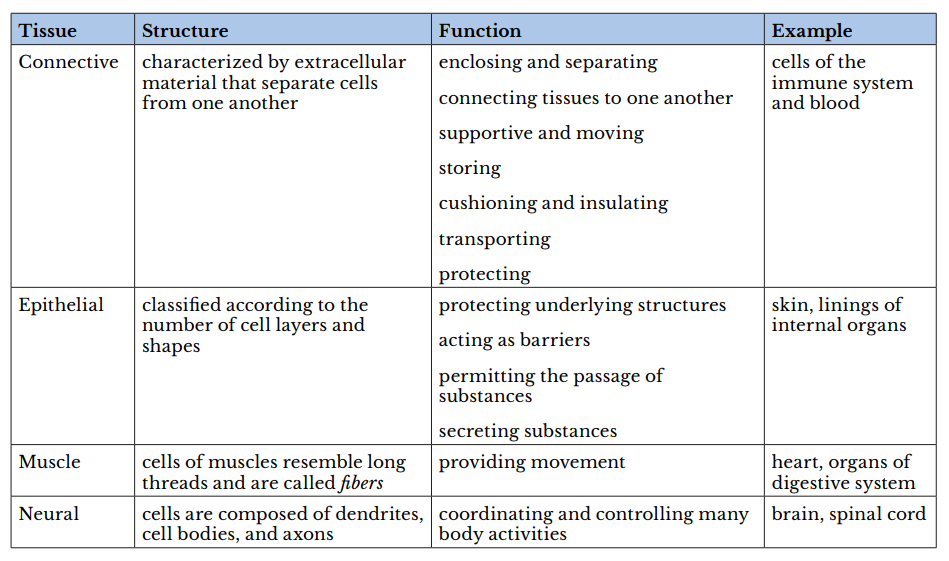
Atissueis a group of cells with similar structure and function and similar extracellular substances located between the cells. The table below describes the four primary tissues found in the human body.
body.
Correct Answer is D
Explanation
The primary function of the respiratory system is to provide oxygen to and remove carbon dioxide from the body. In addition to gas exchange, the respiratory system enables a person to breathe. Breathing, or inhalation, is essential to life. It is the mechanism that provides oxygen to the body. Without oxygen, cells are unable to perform their functions necessary to keep the body alive. The primary muscle of inspiration is the diaphragm. Known as the chest cavity, this dome shaped structure flattens when it contracts. The rib cage moves outward, allowing outside air to be drawn into the lungs. During relaxation, the diaphragm returns to its dome shape and the rib cage moves back to its natural position. This causes the chest cavity to push air out of the lungs.
The respiratory system can be functionally divided into two parts:
- Air-conducting portion: Air is delivered to the lungs. This region consists of the upper and lower respiratory tract—specifically, the larynx, trachea, bronchi, and bronchioles.
- Gas exchange portion: Gas exchange takes place between the air and the blood. This portion includes the lungs, alveoli, and capillaries.
Correct Answer is D
Explanation
Kidneys makes urine isincorrect.Kidneys do not make urine. They help regulate water balance, regulate levels of electrolytes such as sodium and potassium, and eliminate metabolic wastes. Urine is a byproduct of these functions.
As a person ages, kidney tissue and filtration capacity increase isincorrect. As a person ages, the kidneys and bladder change. This can affect functions such as bladder control and how well the kidneys filter blood. Kidney changes range from a decrease in kidney tissue to decreased filtration capacity.
Kidneys help regulate water balance iscorrect.Kidneys help regulate water balance, regulate levels of electrolytes such as sodium and potassium, and eliminate metabolic wastes. Urine is a byproduct of these functions.
Regulates levels of electrolytes such as sodium and potassium iscorrect.There must be a continual balance of water and salt in the blood. The urinary system, specifically the kidneys, help maintain this balance. It also balances levels of metabolites or electrolytes such as sodium, potassium, and calcium.
Eliminates metabolic wastes iscorrect. Urea, creatinine, uric acid, and ammonium are the primary types of nitrogenous wastes excreted from the body. The urinary system also detects and excretes excess water from the blood and out of the body.
Correct Answer is A
Explanation
The cell cycle is an organized process divided into two phases:interphaseand theM (mitotic) phase. During interphase, the cell grows and copies its DNA. After the cell reaches the M phase, division of the two new cells can occur. The G1, S, and G2phases make up interphase.
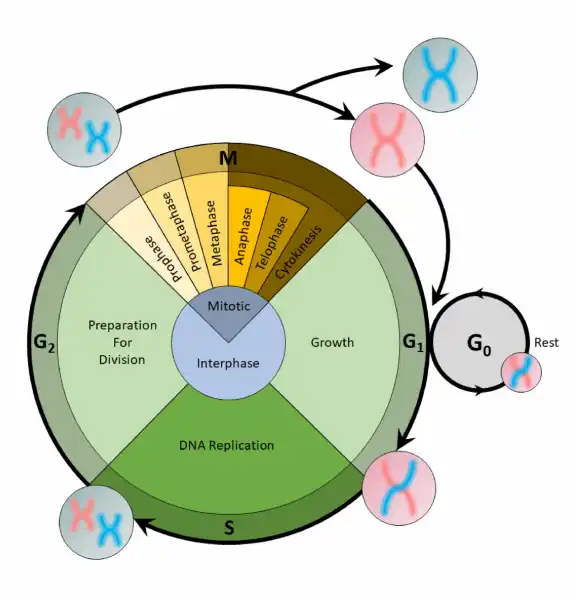
- G1:The first gap phase, during which the cell prepares to copy its DNA
- S:The synthesis phase, during which DNA is copied
- G2:The second gap phase, during which the cell prepares for cell division
It may appear that little is happening in the cell during the gap phases. Most of the activity occurs at the level of enzymes and macromolecules. The cell produces things like nucleotidesfor synthesizing new DNA strands, enzymes for copying the DNA, and tubulin proteins for building the mitotic spindle. During the S phase, the DNA in the cell doubles, but few other signs are obvious under the microscope. All the dramatic events that can be seen under a microscope occur during the M phase: the chromosomes move, and the cell splits into two new cells with identical nuclei.
Correct Answer is B
Explanation
In this reaction, chlorine (Cl2) is an element in the reaction that replaces iodine in the compound sodium iodide (NaI). This allows chlorine to form a compound with sodium (NaCl) and leaves iodine (I2) as an element.
Synthesisreactions involve two or more reactants (A and B) combining to form one product (AB). In the example provided, hydrogen (H2) and oxygen (O2) begin as separate elements. At the end of the reaction, the hydrogen and oxygen atoms are bonded in a molecule of water (H2O).
Decompositionreactions have only one reactant (AB) that breaks apart into two or more products (A and B). In the example above, hydrogen peroxide (H2O2) breaks apart into two smaller molecules: water (H2O) and oxygen (O2).
Single-replacementreactions involve two reactants, one compound (AB) and one element (C). In this type of reaction, one element replaces another to form a new compound (AC), leaving one element by itself (B). In the example, zinc replaces hydrogen in hydrochloric acid (HCl). As a result, zinc forms a compound with chlorine, zinc chloride (ZnCl2), and hydrogen (H2) is left by itself.
Double-replacementreactions involve two reactants, both of which are compounds made of two components (AB and CD). In the example, silver nitrate, composed of silver (Ag1+) and nitrate (NO31-) ions, reacts with sodium chloride, composed of sodium (Na1+) and chloride (Cl1-) ions. The nitrate and chloride ions switch places to produce two compounds that are different from those in the reactants.
Combustionreactions occur when fuels burn, and they involve specific reactants and products, as seen in the examples below. Some form of fuel that contains carbon and hydrogen is required. Examples of such fuels are methane, propane in a gas grill, butane in a lighter, and octane in gasoline. Notice that these fuels all react with oxygen, which is necessary for anything to burn. In all combustion reactions, carbon dioxide, water, and energy are produced. When something burns, energy is released, which can be felt as heat and seen as light.
Correct Answer is C
Explanation
Homeostasisis the existence and maintenance of a relatively constant environment within the body. Each cell of the body is surrounded by a small amount of fluid, and the normal functions of each cell depend on the maintenance of its fluid environment within a narrow range of conditions, including temperature, volume, and chemical content. These conditions are known asvariables. For example, body temperature is a variable that can increase in a hot environment or decrease in a cold environment.
There are two types of feedback mechanisms in the human body: negative and positive.
- Negative Feedback: Most systems of the body are regulated by negative feedback mechanisms, which maintain homeostasis. Negative means that any deviation from the set point is made smaller or is resisted. The maintenance of normal blood pressure is a negative-feedback mechanism. Normal blood pressure is important because it is responsible for moving blood from the heart to tissues.
- Positive Feedback: Positive-feedback mechanisms are not homeostatic and are rare in healthy individuals. Positive means that when a deviation from a normal value occurs, the response of the system is to make the deviation even greater. Positive feedback therefore usually creates a cycle leading away from homeostasis and, in some cases, results in death. Inadequate delivery of blood to cardiac muscle is an example of positive feedback.
Correct Answer is D
Explanation
Because it has more protons than electrons, this atom has a positive charge and can be classified as a cation. When a metal such as sodium reacts to become stable, it loses its valence electrons. At first, it is a neutral atom with 11 protons and 11 electrons. When it loses an electron, the number of protons does not change, and the atom has 11 protons and 10 electrons. Because there is one more positively charged proton, acationforms. A cation is an ion with a net positive charge.
Correct Answer is A
Explanation
This question was extracted from the actual TEAS Exam. Ace your TEAS exam with the actual TEAS 7 questions, Start your journey with us today
Visit Naxlex, the Most Trusted TEAS TEST Platform With Guaranteed Pass of 90%.
Money back guarantee if you use our service and fail the actual exam. Option of personalised live tutor on your area of weakness.
