Which blood group is a universal donor?
A
B
AB
O
Correct Answer : D
A person can be a universal blood donor or acceptor. A universal blood donor has type O blood, while a universal blood acceptor has type AB blood.
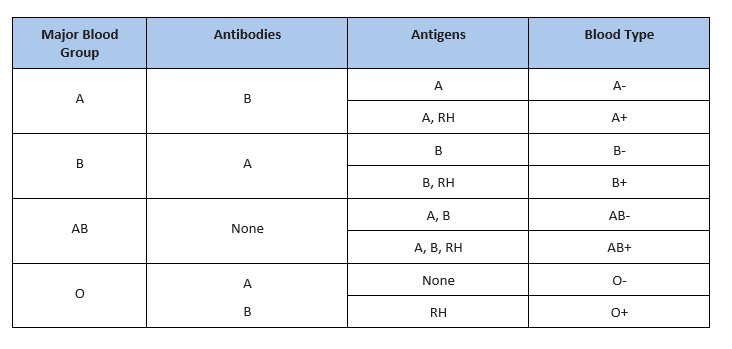
There are several different types or groups of blood, and the major groups are A, B, AB, and O. Blood group is a way to classify blood according to inherited differences of red blood cell antigens found on the surface of a red blood cell. The type of antibody in blood also identifies a particular blood group. Antibodies are proteins found in the plasma. They function as part of the body’s natural defense to recognize foreign substances and alert the immune system.

Depending on which antigen is inherited, parental offspring will have one of the four major blood groups. Collectively, the following major blood groups comprise the ABO system:
- Blood group A: Displays type A antigens on the surface of a red blood cell and contains B antibodies in the plasma.
- Blood group B: Displays type B antigens on the red blood cell’s surface and contains A antibodies in the plasma.
- Blood group O: Does not display A or B antigens on the surface of a red blood cell. Both A and B antibodies are in the plasma.
- Blood group AB: Displays type A and B antigens on the red blood cell’s surface, but neither A nor B antibodies are in the plasma
In addition to antigens, the Rh factor protein may exist on a red blood cell’s surface. Because this protein can be either present (+) or absent (-), it increases the number of major blood groups from four to eight: A+, A-, B+, B-, O+, O-, AB+, and AB-.
TEAS 7 Exam Quiz Bank
HESI A2 Exam Quiz Bank
Find More Questions 📚
Teas 7 Questions: We got the latest updated TEAS 7 questions
100% Money Refund: 100% money back guarantee if you take our full
assessment pass with 80% and fail the actual exam.
Live Tutoring: Fully customized live tutoring lessons.
Guaranteed A Grade: All students who use our services pass with 90%
guarantee.
Related Questions
Correct Answer is D
Explanation
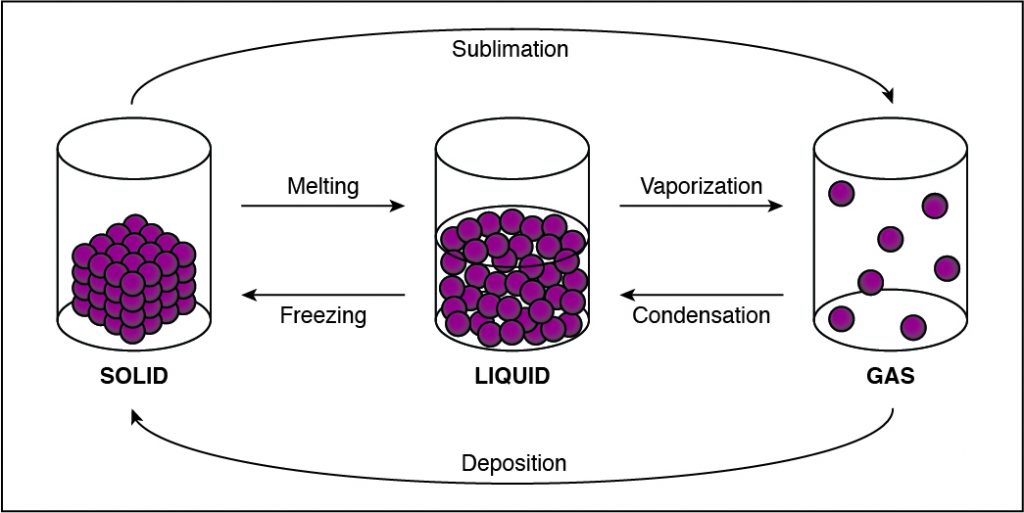
In solids, particles are usually closer together than in other states of matter because of the strong cohesive forces between the particles.
- Solids, liquids, gases, and plasmas differ from one another in the amount of energy that the particles have and the strength of the cohesive forces that hold the particles together.
- Cohesion is the tendency of particles of the same kind to stick to each other.
- A solid has the lowest amount of energy because its particles are packed close together. Liquids have more energy than a solid, and gases have more energy than solids or liquids because the cohesive forces are very weak.
Correct Answer is A
Explanation
Taxonomy is the process of classifying, describing, and naming organisms. There are seven levels in the Linnaean taxonomic system, starting with the broadest level, kingdom, and ending with the species level. For example, in the image the genus level contains two types of bears, but the species level shows one type. Additionally,organisms in each level are found in the level above it. For example, organisms in the order level are part of the class level. This classification system is based on physical similarities across living things. It does not account for molecular or genetic similarities.

Correct Answer is B
Explanation
A control group is a factor that does not change during an experiment. Due to this, it is used as a standard for comparison with variables that do change such as a dependent variable.
Recall that these make up thescientific method,described below:
- Problem:The question created because of an observation.Example: Does the size of a plastic object affect how fast it naturally degrades in a lake?
- Research:Reliable information available about what is observed.Example: Learn how plastics are made and understand the properties of a lake.
- Hypothesis:A predicted solution to the question or problem.Example: If the plastic material is small, then it will degrade faster than a large particle.
- Experiment:A series of tests used to evaluate the hypothesis. Experiments consist of anindependent variablethat the researcher modifies and adependent variablethat changes due to the independent variable. They also include acontrol groupused as a standard to make comparisons.
- Example: Collect plastic particles both onshore and offshore of the lake over time. Determine the size of the particles and describe the lake conditions during this time period.
- Observe:Analyze data collected during an experiment to observe patterns.
- Example: Analyze the differences between the numbers of particles collected in terms of size.
- Conclusion:State whether the hypothesis is rejected or accepted and summarize all results.
- Communicate:Report findings so others can replicate and verify the results.
Correct Answer is B
Explanation
A cell copies its DNA during the S phase, and nucleotides are the building blocks of DNA. Thus, the step preceding the S phase, theG1phase, is the phase of the cell cycle when the cell would contain the most nucleotides.
For a cell to divide into more cells, it must grow, copy its DNA, and produce new daughter cells. Thecell cycleregulates cellular division. This process can either prevent a cell from dividing or trigger it to start dividing.
The cell cycle is an organized process divided into two phases:interphaseand theM (mitotic) phase. During interphase, the cell grows and copies its DNA. After the cell reaches the M phase, division of the two new cells can occur. The G1, S, and G2phases make up interphase.
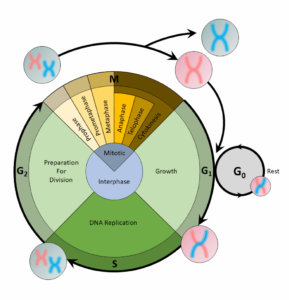
- G1:The first gap phase, during which the cell prepares to copy its DNA
- S:The synthesis phase, during which DNA is copied
- G2:The second gap phase, during which the cell prepares for cell division
It may appear that little is happening in the cell during the gap phases. Most of the activity occurs at the level of enzymes and macromolecules. The cell produces things like nucleotidesfor synthesizing new DNA strands, enzymes for copying the DNA, and tubulin proteins for building the mitotic spindle. During the S phase, the DNA in the cell doubles, but few other signs are obvious under the microscope. All the dramatic events that can be seen under a microscope occur during the M phase: the chromosomes move, and the cell splits into two new cells with identical nuclei.
Correct Answer is D
Explanation
The duodenum is the first part of the small intestines, located between the stomach and the middle part of the small intestines (jejunum). Once food has mixed with acid in the stomach, it moves into the duodenum, where it then mixes with bile from the gallbladder and digestive juices secreted from the pancreas. In the duodenum, absorption of vitamins, minerals, and nutrients begins.
Correct Answer is D
Explanation
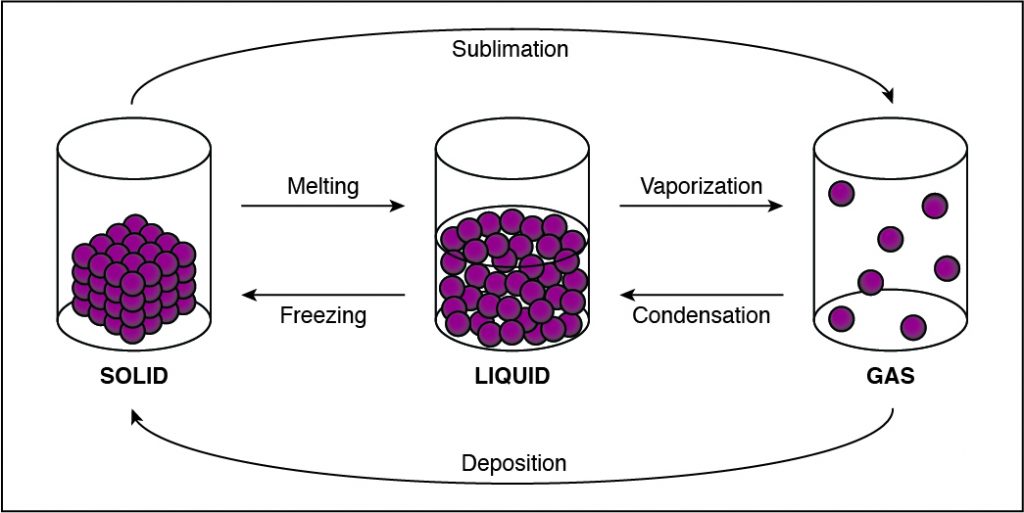
If the cohesion between particles decreases, then the particles must be undergoing a phase change that allows particles to move farther apart. This happens when a substance vaporizes and turns from liquid to gas. Any phase change that moves to the right in the diagram above requires energy to be added to the system because the substance has more energy at the end of the phase change. The phase changes aremelting,vaporization (boiling), andsublimation. When energy is added, particles move faster and can break away from each other more easily as they move to a state of matter with a higher amount of energy. This is most commonly done by heating the substance.
Correct Answer is D
Explanation
The primary function of the respiratory system is to provide oxygen to and remove carbon dioxide from the body. In addition to gas exchange, the respiratory system enables a person to breathe. Breathing, or inhalation, is essential to life. It is the mechanism that provides oxygen to the body. Without oxygen, cells are unable to perform their functions necessary to keep the body alive. The primary muscle of inspiration is the diaphragm. Known as the chest cavity, this dome shaped structure flattens when it contracts. The rib cage moves outward, allowing outside air to be drawn into the lungs. During relaxation, the diaphragm returns to its dome shape and the rib cage moves back to its natural position. This causes the chest cavity to push air out of the lungs.
The respiratory system can be functionally divided into two parts:
- Air-conducting portion: Air is delivered to the lungs. This region consists of the upper and lower respiratory tract—specifically, the larynx, trachea, bronchi, and bronchioles.
- Gas exchange portion: Gas exchange takes place between the air and the blood. This portion includes the lungs, alveoli, and capillaries.
Correct Answer is B
Explanation
The number of protons, 28, gives the atomic number, which identifies this atom as nickel. The mass is the number after the dash in the isotope name, which is determined by adding the numbers of protons and neutrons (28 + 32 = 60).
Correct Answer is D
Explanation
Because this is a positive correlation, if the nutrient concentration increases or decreases, plant height will either increase or decrease accordingly.
While analyzing data, scientists tend to observe cause-and-effect relationships. These relationships can be quantified using correlations.Correlationsmeasure the amount of linear association between two variables. There are three types of correlations:
Positive correlation:
As one variable increases, the other variable also increases. This is also known as a direct correlation.
Negative correlation:
As one variable increases, the other decreases. The opposite is true if one variable decreases. A negative correlation is also known as an inverse correlation or an indirect correlation.
No correlation:
There is no connection or relationship between two variables.
Correct Answer is B
Explanation
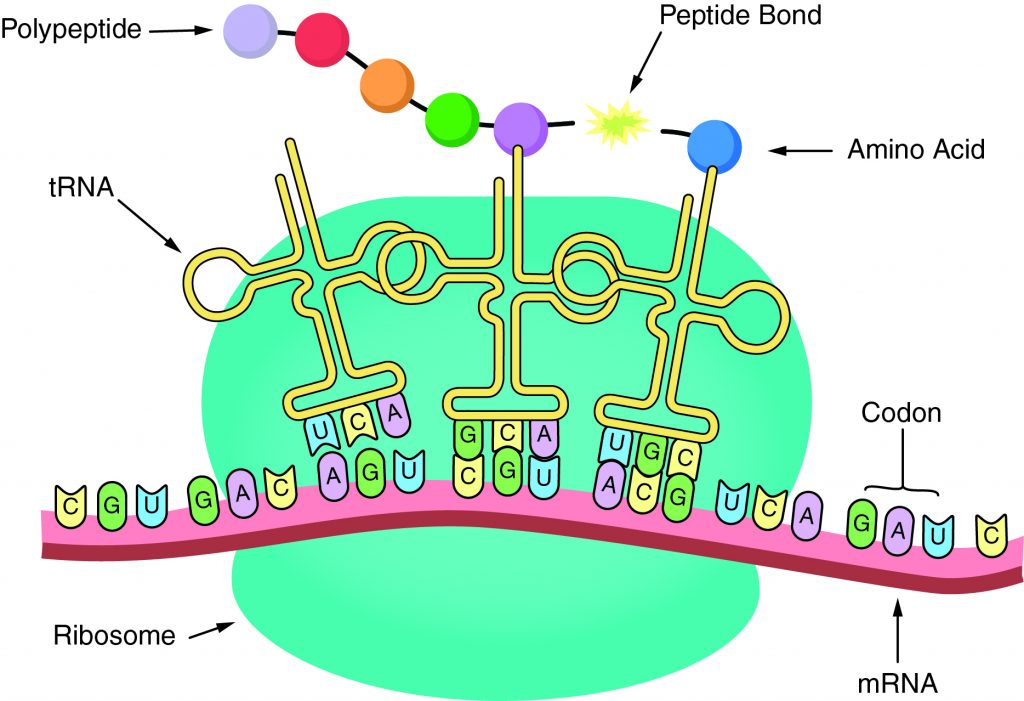
The sequence of amino acids in a gene determines the primary structure of a protein. The components necessary fortranslationare located in the cytoplasm. Translation is the making of proteins by mRNA binding to a ribosome with the start codon that initiates the production of amino acids. Apeptide bondforms and connects the amino acids together. The sequence of amino acids determines the protein’s structure, which determines its function.
This question was extracted from the actual TEAS Exam. Ace your TEAS exam with the actual TEAS 7 questions, Start your journey with us today
Visit Naxlex, the Most Trusted TEAS TEST Platform With Guaranteed Pass of 90%.
Money back guarantee if you use our service and fail the actual exam. Option of personalised live tutor on your area of weakness.
