Which of the following structures is present in both prokaryotic and eukaryotic cells?
Cell membrane
Golgi apparatus
Chloroplasts
Endoplasmic reticulum
Correct Answer : A
The cell membrane is present in both prokaryotic and eukaryotic cells.
The cell membrane is a thin, flexible barrier that surrounds all cells and separates the inside of the cell from the outside environment.
It is composed of a lipid bilayer and regulates the movement of substances into and out of the cell.
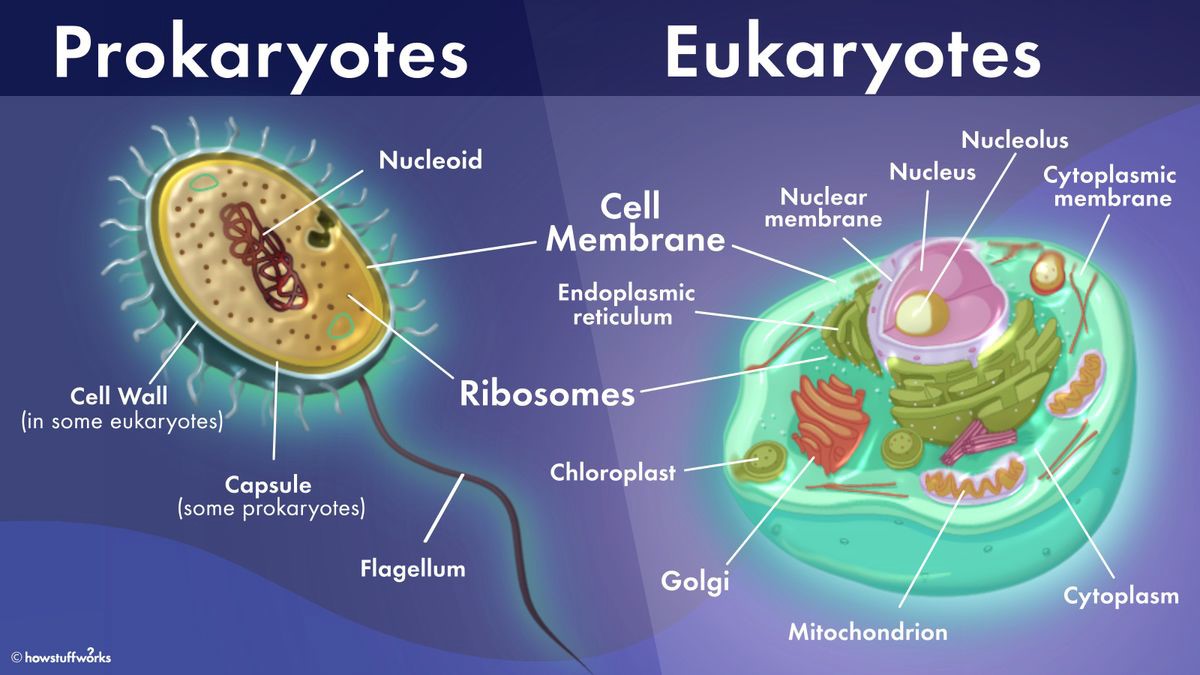
Choice B is incorrect because the Golgi apparatus is not present in prokaryotic cells.
The Golgi apparatus is an organelle found in eukaryotic cells that is involved in modifying, sorting, and packaging proteins and lipids for transport to other parts of the cell or to be secreted outside the cell.
Choice C is incorrect because chloroplasts are not present in prokaryotic cells.
Chloroplasts are organelles found in plant cells and some algae that are responsible for photosynthesis.
Choice D is incorrect because the endoplasmic reticulum is not present in prokaryotic cells.
The endoplasmic reticulum is an organelle found in eukaryotic cells that is involved in protein synthesis and lipid metabolism.
TEAS 7 Exam Quiz Bank
HESI A2 Exam Quiz Bank
Find More Questions 📚
Teas 7 Questions: We got the latest updated TEAS 7 questions
100% Money Refund: 100% money back guarantee if you take our full
assessment pass with 80% and fail the actual exam.
Live Tutoring: Fully customized live tutoring lessons.
Guaranteed A Grade: All students who use our services pass with 90%
guarantee.
Related Questions
Correct Answer is A
Explanation
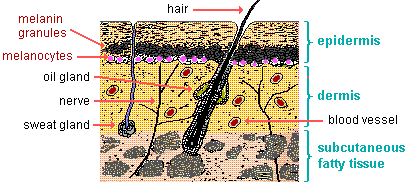
Melanin.
Melanin is a pigment produced by cells called melanocytes in the skin.
It protects the skin from ultraviolet (UV) radiation by absorbing and dissipating over 99.9% of absorbed UV radiation.
This helps to prevent DNA damage and other adverse effects of UV radiation on the skin.
Choice B.
Perspiration is not correct because it is a fluid produced by sweat glands in the skin that helps to regulate body temperature, but it does not protect the skin from UV radiation.
Choice C.
Sebum is not correct because it is an oily substance produced by sebaceous glands in the skin that helps to lubricate and protect the skin, but it does not protect the skin from UV radiation.
Choice D.
Keratin is not correct because it is a fibrous protein that provides strength and durability to the skin, hair and nails, but it does not protect the skin from UV radiation.
Correct Answer is C
Explanation
Ovulation is the process in which an ovarian follicle matures and releases a reproductive egg.
During ovulation, a mature egg is released from the female ovary, enabling it to be fertilized by male sperm cells 1.
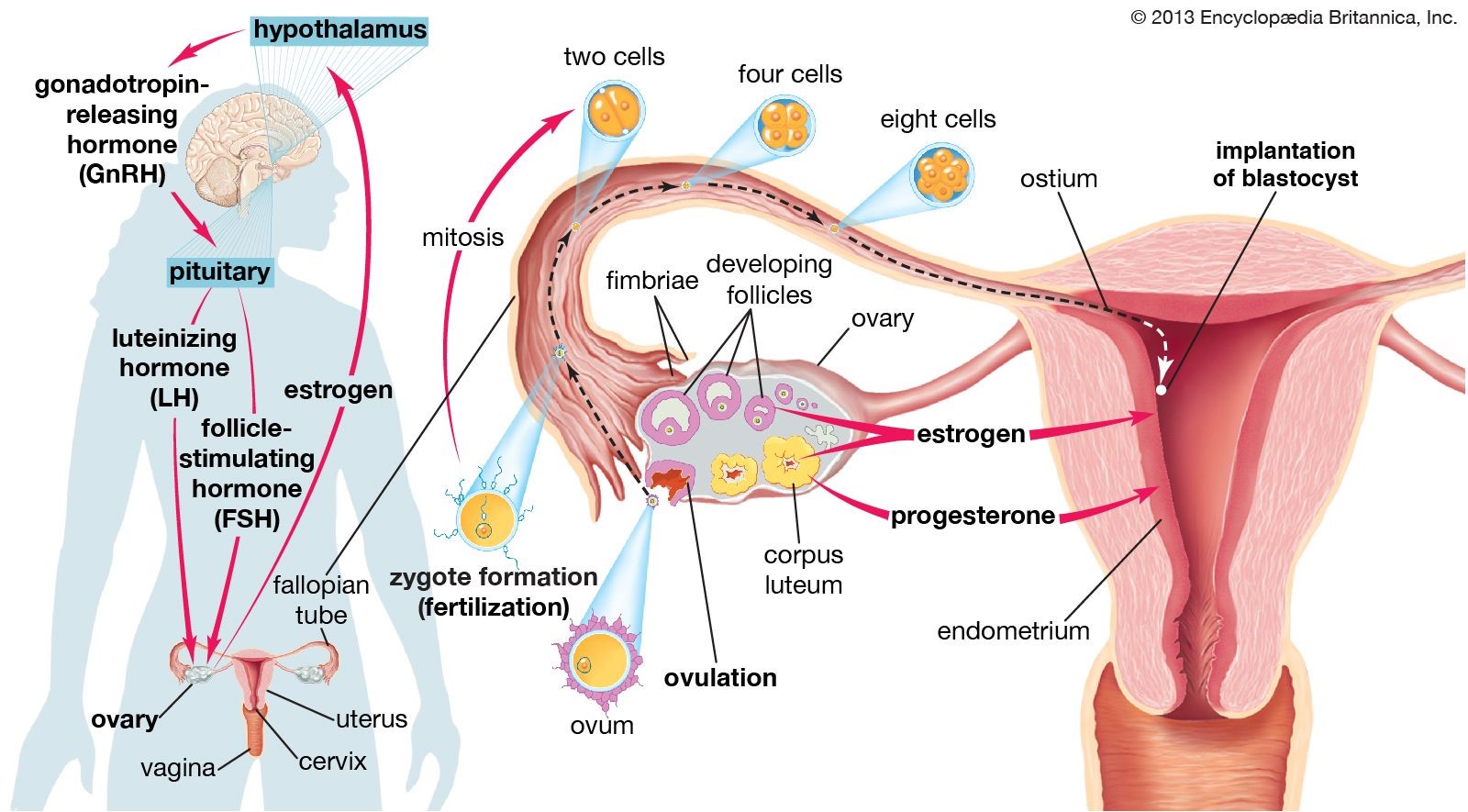
Choice A is incorrect because menstruation is the process of shedding the uterine lining, which occurs when an egg is not fertilized.
Choice B is incorrect because fertilization is the process of a sperm cell joining with an egg cell to form a zygote.
Choice D is incorrect because oogenesis is the process of forming female gametes (eggs) in the ovaries.
Correct Answer is D
Explanation
The cytoskeleton of a cell is comprised of protein fibers that provide structural support and help maintain the shape of the cell.
These protein fibers include microfilaments, intermediate filaments, and microtubules.
Choice A.
Carbohydrates is not the correct answer because carbohydrates are a type of macromolecule that provides energy to cells and are not a component of the cytoskeleton.
Choice B.
Nucleic acids is not the correct answer because nucleic acids are macromolecules that store and transmit genetic information and are not a component of the cytoskeleton.
Choice C.
Lipids is not the correct answer because lipids are a type of macromolecule that makes up cell membranes and are not a component of the cytoskeleton.
Correct Answer is B
Explanation
The atomic number of an atom is equal to the number of protons in its nucleus.
In this case, the atom has 12 protons, so its atomic number is 12.
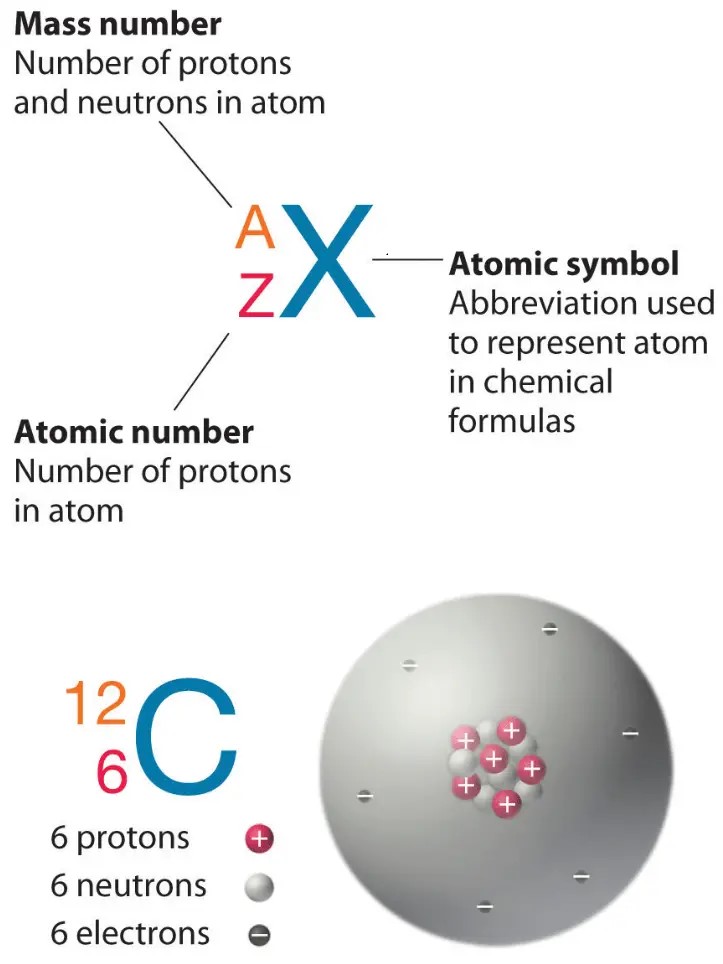
Choice A, 24, is not the correct answer because it represents the sum of the number of protons and neutrons in the atom’s nucleus, which is known as the mass number.
Choice C, 1, is not the correct answer because it does not represent the number of protons in the atom’s nucleus.
Choice D, 144, is not the correct answer because it represents the square of the mass number and does not represent any property of the atom.
Correct Answer is B
Explanation
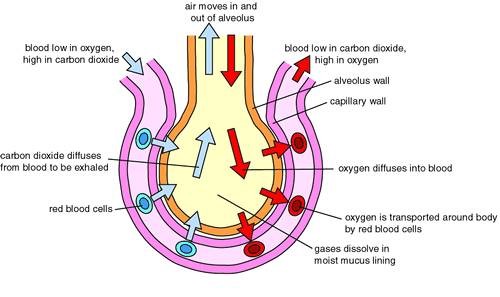
Diffusion down a concentration gradient causes most of the carbon dioxide from the blood to move into the alveoli.
The alveoli are tiny air sacs in the lungs where gas exchange occurs.
Carbon dioxide is a waste product of cellular respiration and is carried by the blood to the lungs to be exhaled.
In the lungs, carbon dioxide diffuses from the blood (where its concentration is high) into the alveoli (where its concentration is lower) down its concentration gradient.
Choice A is incorrect because carbon dioxide is not converted to carbon monoxide in the body.
Choice C is incorrect because passive transport using carrier proteins is not the primary mechanism by which carbon dioxide moves from the blood into the alveoli.
Choice D is incorrect because active transport using energy is not involved in the movement of carbon dioxide from the blood into the alveoli.
Correct Answer is D
Explanation
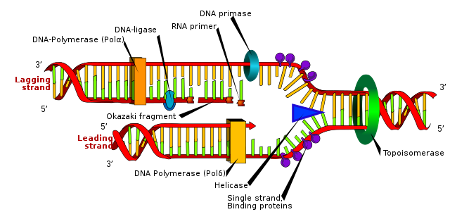
Genes are used in the process of DNA sequencing to determine the order of nucleotides in a DNA molecule.
Choice B.
Blood types is not the correct answer because blood types are determined by the presence or absence of specific antigens on the surface of red blood cells and are not directly related to DNA sequencing.
Choice C.
Hormones is not the correct answer because hormones are chemical messengers produced by glands in the body and are not directly involved in DNA sequencing.
Choice D.
Genes is the correct answer because genes are used in the process of DNA sequencing to determine the order of nucleotides in a DNA molecule.
Correct Answer is D
Explanation
A totipotent cellcan self-renew by dividing and develop into the three primary germ cell layers of the early embryo and into extra-embryonic tissues such as the placenta.
A fertilized egg is a totipotent stem cell and as such can develop into any specialized cell found in the organism.
Choice A is not correct because totipotent cells do not fight infectious diseases.
Choice B is not correct because totipotent cells do not aid in the maturation of sex cells.
Choice C is not correct because totipotent cells do not carry electrical impulses.
Correct Answer is B
Explanation
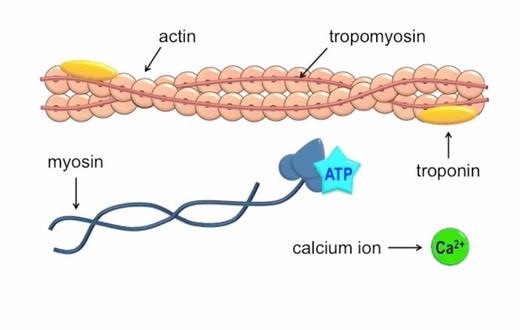
Calcium ions play a crucial role in initiating muscle contraction.
When a muscle cell is stimulated to contract by an action potential, calcium channels open in the sarcoplasmic membrane and release calcium into the sarcoplasm.
Some of this calcium attaches to troponin, which causes it to change shape.
This shape change exposes binding sites for myosin on the actin filaments.
Myosin’s binding to actin causes crossbridge formation, and contraction of the muscle begins.
The other ions mentioned in the question do not have this specific role in muscle contraction.
Potassium ions are important for maintaining the resting membrane potential of cells, but they do not bind to the troponin complex.
Phosphorus ions are important for energy metabolism, but they do not bind to the troponin complex.
Sodium ions are important for generating action potentials, but they do not bind to the troponin complex.
Correct Answer is C
Explanation
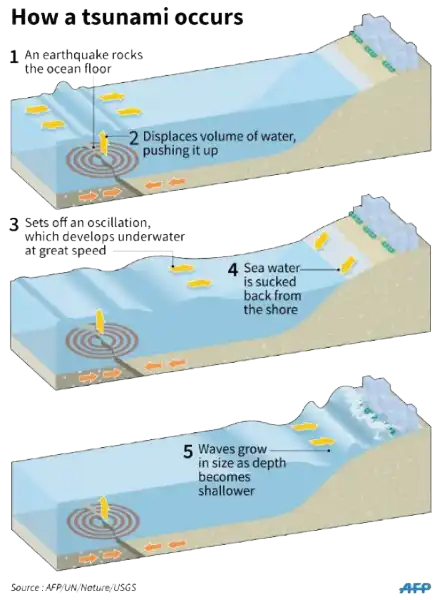
A tsunami is a catastrophic ocean wave that is usually caused by a submarine earthquake.
It can also be caused by an underwater or coastal landslide, the eruption of a volcano, or the impact of a meteor or comet in a body of water.
Choice A is not correct because sunspot activity does not cause tsunamis.
Choice B is not correct because lightning strikes do not cause tsunamis.
Choice D is not correct because flooding does not cause tsunamis.
Correct Answer is D
Explanation
Triple covalent bonds.
Nitrogen gas (N2) is an extremely stable molecule because it consists of two nitrogen atoms bonded together by a triple covalent bond.
A covalent bond is a type of chemical bond where atoms share electrons to form a molecule.
In a triple covalent bond, three pairs of electrons are shared between the two atoms, resulting in a very strong bond that makes the molecule extremely stable.
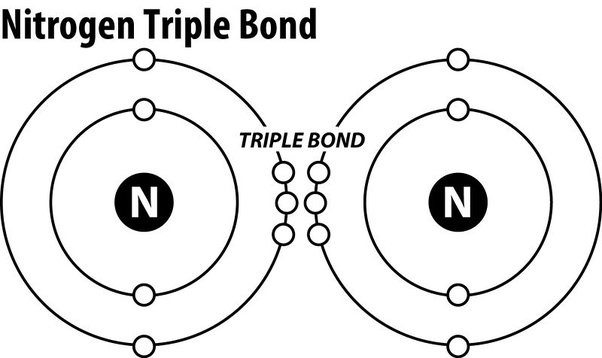
Choice A.
Ionic bonds is not correct because ionic bonds involve the transfer of electrons from one atom to another to form ions, which are then attracted to each other due to their opposite charges.
Nitrogen gas does not contain ions and is not held together by ionic bonds.
Choice B.
Hydrogen bonds is not correct because hydrogen bonds are weak electrostatic attractions between molecules that contain hydrogen atoms bonded to highly electronegative atoms such as oxygen or nitrogen.
Nitrogen gas does not contain hydrogen atoms and is not held together by hydrogen bonds.
Choice C.
Resonance bonds is not correct because resonance refers to the delocalization of electrons in a molecule where multiple Lewis structures can be drawn to represent the molecule.
Nitrogen gas has a single Lewis structure and does not exhibit resonance.
This question was extracted from the actual TEAS Exam. Ace your TEAS exam with the actual TEAS 7 questions, Start your journey with us today
Visit Naxlex, the Most Trusted TEAS TEST Platform With Guaranteed Pass of 90%.
Money back guarantee if you use our service and fail the actual exam. Option of personalised live tutor on your area of weakness.
