A researcher collects data on the number of cars passing through a busy intersection at different times of the day for a month. This data would be most useful to analyze which of the following:
traffic patterns during rush hour
pedestrian movement during the day
air pollution levels in the area
noise levels in the area
The Correct Answer is A
The data collected by the researcher on the number of cars passing through a busy intersection at different times of the day for a month would be most useful for analyzing traffic patterns during rush hour.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is A
Explanation
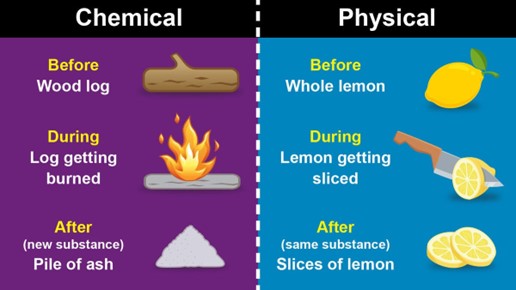
A physical change is a change that affects the physical properties of a substance but does not change its chemical identity. Physical changes include changes in state, such as melting or boiling, changes in shape or size, and changes in phase, such as the dissolution of a solid in a liquid. In a physical change, the atoms and molecules of the substance are rearranged, but no new substances are formed.
A chemical change, on the other hand, is a change that results in the formation of new substances with different chemical properties. Chemical changes involve the breaking of chemical bonds between atoms and the formation of new bonds to create new compounds. Chemical changes are usually accompanied by a change in color, the formation of a gas or a solid, or the release or absorption of energy.
Overall, the main difference between a physical change and a chemical change is that a physical change only affects the physical properties of a substance while a chemical change results in the formation of new substances with different chemical properties.
Correct Answer is C
Explanation
The molecular geometry of a molecule of Sulphur dioxide (SO2) is bent or V-shaped. This is because of the presence of two lone pairs on the sulfur atom, which cause repulsion and distort the bond angles in the molecule.
SO2 has a central sulfur atom bonded to two oxygen atoms by double bonds. The two double bonds and the two lone pairs of electrons on the sulfur result in a trigonal planar arrangement of electron pairs around the sulfur atom. However, the repulsion between the lone pairs causes the two oxygen atoms to be pulled closer together, resulting in a bent or V-shaped molecular geometry.
The bent molecular geometry of SO2 affects its properties, such as its polarity and reactivity. SO2 is a polar molecule due to the asymmetric distribution of electrons, which results in a partial positive charge on the sulfur atom and partial negative charges on the oxygen atoms. This polarity makes SO2 a good solvent and reactant in chemical reactions, as well as a contributor to air pollution and acid rain.

Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
