How many times stronger is the hydrogen-ion concentration of a pH 4 solution as compared with a pH 9 solution?
0.00001
5
100,000
50
The Correct Answer is C
The correct answer is c. 100,000. The pH scale is a logarithmic scale, which means that each change of one pH unit represents a tenfold change in the hydrogen-ion concentration. A pH 4 solution has a hydrogen-ion concentration that is 10^5 (or 100,000) times greater than that of a pH 9 solution.
A. 0.00001 is the hydrogen-ion concentration of a pH 9 solution as compared with a pH 4 solution.
B. 5 is the difference in pH units between a pH 4 solution and a pH 9 solution.
D. 50 is not the correct answer.

Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is A
Explanation
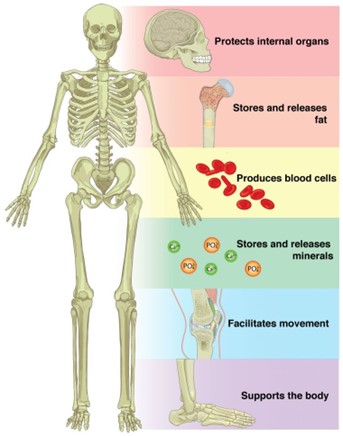
Storage of minerals. Bones serve as a storage site for minerals such as calcium and phosphorus. These minerals are essential for various bodily functions and can be released from the bones into the bloodstream when needed.
B. Detoxification of alcohol is not a function of bone. This process occurs primarily in the liver.
C. Secretion of hormones is not a function of bone. Hormones are produced and secreted by glands such as the pituitary gland, thyroid gland, and adrenal glands.
D. Production of otoliths is not a function of bone. Otoliths are small calcium carbonate structures found in the inner ear of fish and other vertebrates that help with balance and hearing.
Correct Answer is D
Explanation
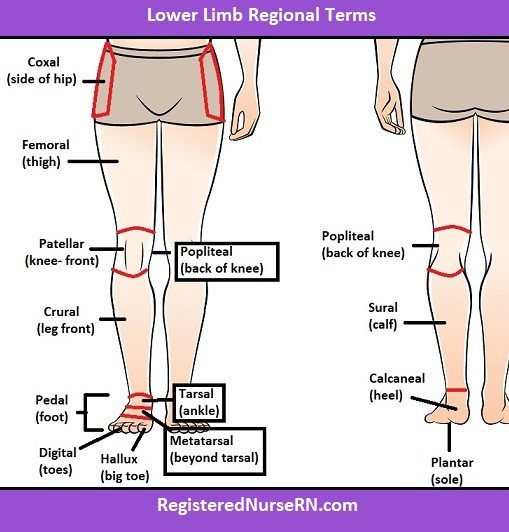
The tibia and fibula are located in the crural region of the body, which is the lower leg between the knee and ankle. The coxal region refers to the hip area, the antecubital region is the front of the elbow, and the tarsal region is the ankle and foot
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
