Which of the following is an example of a double-blind study?
Participants are randomly assigned to a treatment group or a control group
Participants and researchers both know which group participants are assigned to
Participants do not know which group they are assigned to, but researchers do
Both participants and researchers do not know which group participants are assigned to
The Correct Answer is D
A double-blind study is a research design in which neither the participants nor the researchers know which group participants are assigned to. This is done to minimize bias and ensure that the results of the study are as objective as possible. In a double-blind study, the treatment and control groups are randomly assigned, and the participants and researchers are unaware of which group each participant is assigned to. Option a) is an example of a randomized controlled trial, which is a common research design, but it is not necessarily double-blind. Option b) is an example of an open-label study, in which both the participants and the researchers know which group each participant is assigned to. Option c) is an example of a single-blind study, in which the participants do not know which group they are assigned to, but the researchers do.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is A
Explanation
The data collected by the researcher on the number of cars passing through a busy intersection at different times of the day for a month would be most useful for analyzing traffic patterns during rush hour.
Correct Answer is B
Explanation
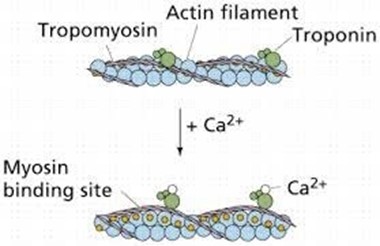
Muscle contraction is a complex process that involves the interaction between actin and myosin filaments in the muscle fibers. The sliding of these filaments is initiated by the release of calcium ions from the sarcoplasmic reticulum, a specialized organelle in muscle cells. The calcium ions bind to the protein troponin, which causes a conformational change in the troponin-tropomyosin complex, exposing the myosin-binding sites on actin. This allows the myosin heads to bind to actin, forming cross-bridges that pull the actin filaments toward the center of the sarcomere, resulting in muscle contraction.
Option a) is incorrect because calcium does not bind to tropomyosin directly, but rather binds to the protein troponin, causing a conformational change in the troponin-tropomyosin complex.
Option c) is incorrect because calcium does not activate motor neurons, but rather is released from the sarcoplasmic reticulum in response to an action potential that travels down the motor neuron to the neuromuscular junction.
Option d) is incorrect because calcium is required for muscle contraction, not relaxation. The relaxation of muscles after contraction is due to the active transport of calcium ions back into the sarcoplasmic reticulum, which allows the troponin-tropomyosin complex to return to its resting conformation, blocking the myosin-binding sites on actin and ending the cross-bridge cycle.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
