What are the five regions of the vertebral column, starting from the top and moving downwards?
Cervical, thoracic, lumbar, sacral, coccygeal
Thoracic, cervical, lumbar, sacral, coccygeal
Lumbar, thoracic, cervical, coccygeal, sacral
Sacral, lumbar, cervical, thoracic, coccygeal
Correct Answer : A
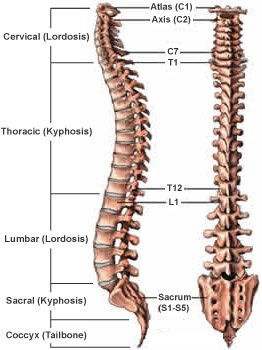
The vertebral column, also known as the spine or spinal column, is a series of bones called vertebrae that extend from the skull to the pelvis. It provides support for the body and protects the spinal cord. The five regions of the vertebral column, starting from the top and moving downwards, are:
- Cervical: This region is made up of seven vertebrae and is located in the neck. The first two cervical vertebrae, the atlas and the axis, are specialized to allow for head movement.
2. Thoracic: This region is made up of twelve vertebrae and is located in the upper and middle back. The thoracic vertebrae are larger than the cervical vertebrae and articulate with the ribs.
3. Lumbar: This region is made up of five vertebrae and is located in the lower back. The lumbar vertebrae are the largest and strongest of the vertebrae.
4. Sacral: This region is made up of five fused vertebrae and is located in the pelvis. The sacrum forms the posterior wall of the pelvis and articulates with the hip bones.
5. Coccygeal: This region is made up of four fused vertebrae and is located at the base of the vertebral column. The coccyx, or tailbone, provides atachment points for muscles and ligaments.
 |
TEAS 7 Exam Quiz Bank
HESI A2 Exam Quiz Bank
Find More Questions 📚
Teas 7 Questions: We got the latest updated TEAS 7 questions
100% Money Refund: 100% money back guarantee if you take our full
assessment pass with 80% and fail the actual exam.
Live Tutoring: Fully customized live tutoring lessons.
Guaranteed A Grade: All students who use our services pass with 90%
guarantee.
Related Questions
Correct Answer is D
Explanation
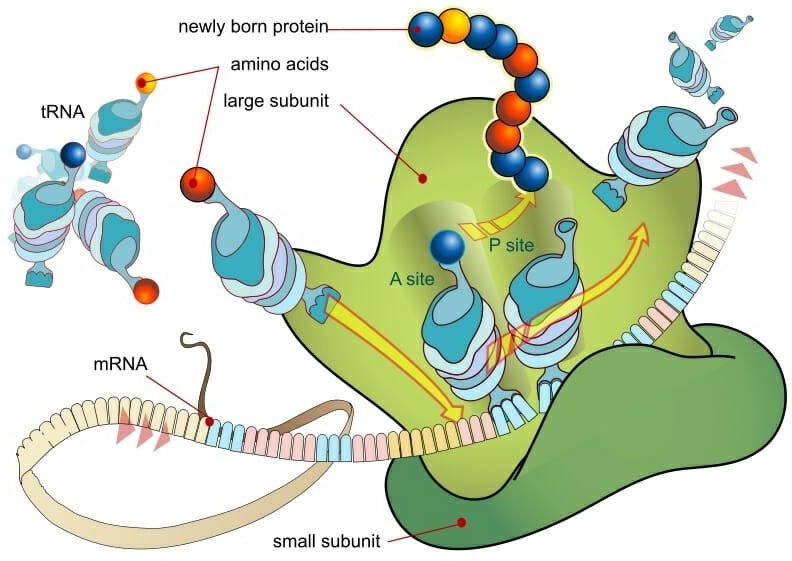
Ribosomes are small, spherical structures found in all living cells, including bacteria, archaea, and eukaryotes. Their primary function is to synthesize proteins using the genetic information stored in the cell's DNA. Ribosomes are composed of two subunits, one large and one small, that come together during protein synthesis.
Ribosomes read the genetic information stored in mRNA (messenger RNA) and use this information to assemble amino acids in the correct order to form a protein. The ribosome moves along the mRNA, adding one amino acid at a time to the growing protein chain until it reaches the end of the mRNA and the protein is complete.
Proteins are essential for a wide variety of cellular functions, including catalyzing chemical reactions, providing structural support, and transporting molecules across cell membranes. Therefore, ribosomes play a critical role in the overall function and survival of a cell.
Correct Answer is C
Explanation
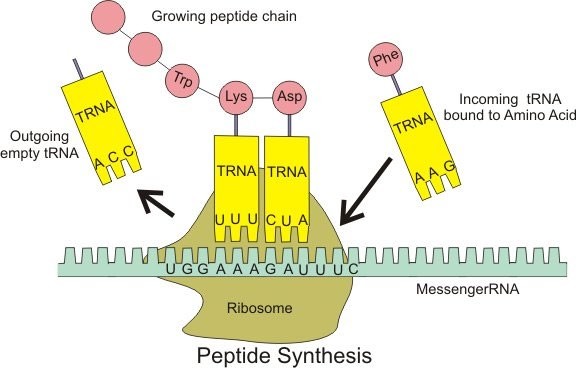
Transfer RNA (tRNA) is a type of RNA molecule that carries amino acids to the ribosome during protein synthesis. Each tRNA molecule has a specific sequence of three nucleotides called an anticodon, which pairs with a complementary codon in the messenger RNA (mRNA) sequence. Each tRNA also carries a specific amino acid that corresponds to the codon it recognizes, allowing the ribosome to link the amino acids together in the correct order to form a protein.
In contrast, messenger RNA (mRNA) carries the genetic information from the DNA to the ribosome, where it serves as a template for protein synthesis. Ribosomal RNA (rRNA) is a component of the ribosome itself, where it helps to catalyze the formation of peptide bonds between amino acids. Small nuclear RNA (snRNA) is involved in splicing of pre-mRNA molecules during post-transcriptional processing.
 |
Correct Answer is B
Explanation
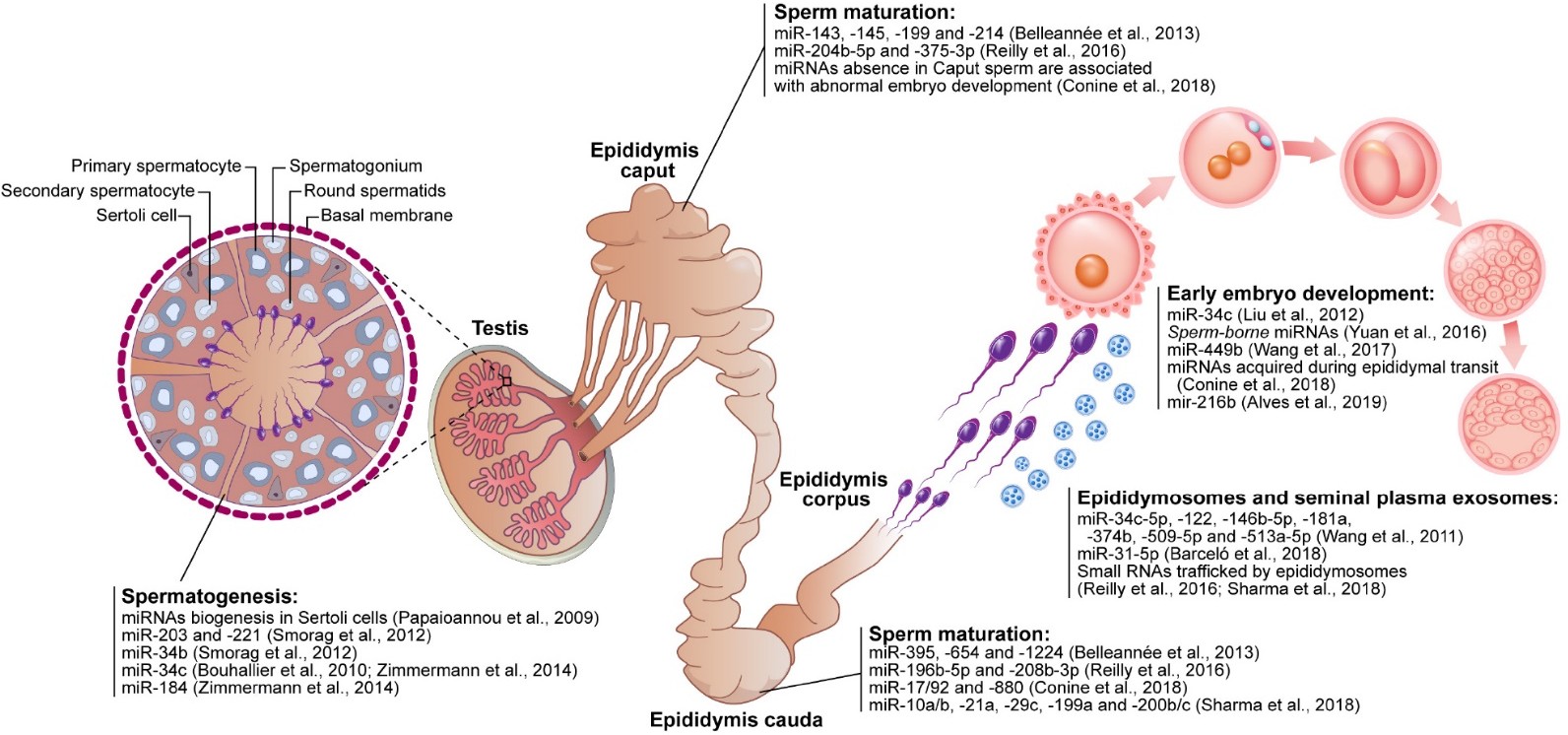
The epididymis is a coiled tube located at the back of each testicle where the sperm mature and are stored until ejaculation. Sperm are produced in the testes and then transported to the epididymis where they undergo maturation and become motile. The epididymis provides a protective environment for the sperm, allowing them to mature and become more resilient to external stressors. During ejaculation, the sperm are transported from the epididymis to the vas deferens and then to the urethra for ejaculation.
 |
Correct Answer is B
Explanation
- Frontal Plane (Coronal Plane) → Divides the body into front (anterior) and back (posterior) halves. ❌
- Transverse Plane → A horizontal plane that divides the body into upper (superior) and lower (inferior) halves. ✅
- Sagittal Plane → Divides the body into left and right halves. ❌
- Coronal Plane → Another name for the Frontal Plane, which divides the body into front and back. ❌
Correct Answer is A
Explanation
The main difference between a solid and a liquid is their physical state and the way their particles are arranged. In a solid, the particles are tightly packed together and have a fixed position, which gives the solid a definite shape and volume. Solids are also characterized by their high density, low compressibility, and high thermal conductivity.
In contrast, the particles in a liquid are more loosely packed and can move around each other, which allows the liquid to take the shape of its container. Liquids have a definite volume but no fixed shape, which means they can be poured or spilled. Liquids also have a lower density than solids, are more compressible than solids, and have lower thermal conductivity than solids.
Option b) is incorrect because it describes the properties of a gas, not a liquid. Option c) is incorrect because solids and liquids have different physical properties. Option d) is incorrect because it describes the properties of a gas, not a liquid or a solid.
 |
Correct Answer is D
Explanation
A double-blind study is a research design in which neither the participants nor the researchers know which group participants are assigned to. This is done to minimize bias and ensure that the results of the study are as objective as possible. In a double-blind study, the treatment and control groups are randomly assigned, and the participants and researchers are unaware of which group each participant is assigned to.
Option a) is an example of a randomized controlled trial, which is a common research design, but it is not necessarily double-blind.
Option b) is an example of an open-label study, in which both the participants and the researchers know which group each participant is assigned to.
Option c) is an example of a single-blind study, in which the participants do not know which group they are assigned to, but the researchers do.

Correct Answer is C
Explanation
Innate immunity is a fundamental aspect of the body's defense mechanism that operates from birth. It offers immediate protection against a wide range of pathogens, such as bacteria, viruses, and fungi, without requiring prior exposure to these invaders.
This defense system is non-specific, meaning it doesn't target a particular pathogen but rather provides a generalized response to various threats. Innate immunity includes physical barriers like the skin and mucous membranes, as well as cellular components such as phagocytes and natural killer cells. These elements work together to detect and neutralize potential threats swiftly, preventing infections from taking hold in the body.
Correct Answer is C
Explanation
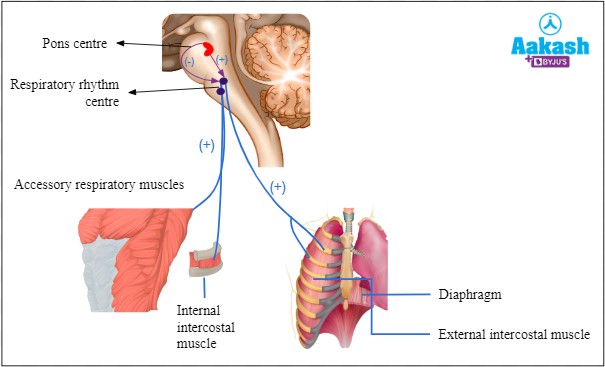
The diaphragm is responsible for regulating breathing rate and depth. It is a dome-shaped muscle located at the bottom of the chest cavity that contracts and relaxes to help move air in and out of the lungs.
Correct Answer is B
Explanation
- A. Sarcoplasmic Reticulum → A specialized endoplasmic reticulum in muscle cells that stores and releases calcium for muscle contraction. ❌
- B. Muscle Fiber → Another name for a muscle cell, which is a long, cylindrical, multinucleated cell that makes up muscle tissue. ✅
- C. Sarcolemma → The cell membrane of a muscle fiber, which surrounds the muscle cell. ❌
- D. Myofilament → Protein structures (actin and myosin) within a muscle fiber that are responsible for contraction. ❌
Correct Answer is D
Explanation
The largest organ in the human body by surface area is the skin. It covers the enre external surface of the body and has an average surface area of about 20 square feet in adults.
This question was extracted from the actual TEAS Exam. Ace your TEAS exam with the actual TEAS 7 questions, Start your journey with us today
Visit Naxlex, the Most Trusted TEAS TEST Platform With Guaranteed Pass of 90%.
Money back guarantee if you use our service and fail the actual exam. Option of personalised live tutor on your area of weakness.
