After soaking for one hour in a solution of unknown concentration, a slice of potato appears to be very soft and limp. You can determine that the solution is
Hypertonic
Hypotonic
Isotonic
Tonic
The Correct Answer is A
Choice A rationale: A hypertonic solution has a higher solute concentration than the potato cell, which means it has a lower water potential. Water will move out of the potato cell by osmosis, causing it to shrink and become soft and limp.
Choice B rationale: A hypotonic solution has a lower solute concentration than the potato cell, which means it has a higher water potential. Water will move into the potato cell by osmosis, causing it to swell and become turgid and firm.
Choice C rationale: An isotonic solution has the same solute concentration as the potato cell, which means it has the same water potential. Water will move in and out of the potato cell at the same rate, causing it to remain unchanged in size and shape.
Choice D rationale: Tonic is not a valid term to describe the solute concentration of a solution. The correct terms are hypertonic, hypotonic, or isotonic.
Choice E rationale: I cannot determine anything without comparing multiple solutions is incorrect because the appearance of the potato slice after soaking in the solution provides enough information to determine the relative solute concentration of the solution.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is B
Explanation
Tonicity is the ability of a solution to cause water movement across a semi-permeable membrane, such as the cell membrane. In the lab, tonicity can be tested by observing the effects of different solutions on cells, such as red blood cells or plant cells. However, the items that have been contaminated with blood, such as slides, pipettes, or gloves, cannot be thrown away in the regular trash can, because they pose a biohazard risk. Biohazardous waste is any material that contains or has been exposed to infectious agents, such as blood, bacteria, viruses, or parasites. Biohazardous waste must be handled and disposed of according to specific safety protocols, such as using autoclaves, incinerators, or biohazard bags. Therefore, after testing tonicity in the lab, the items that have been contaminated with blood cannot be disposed of in the trash can.
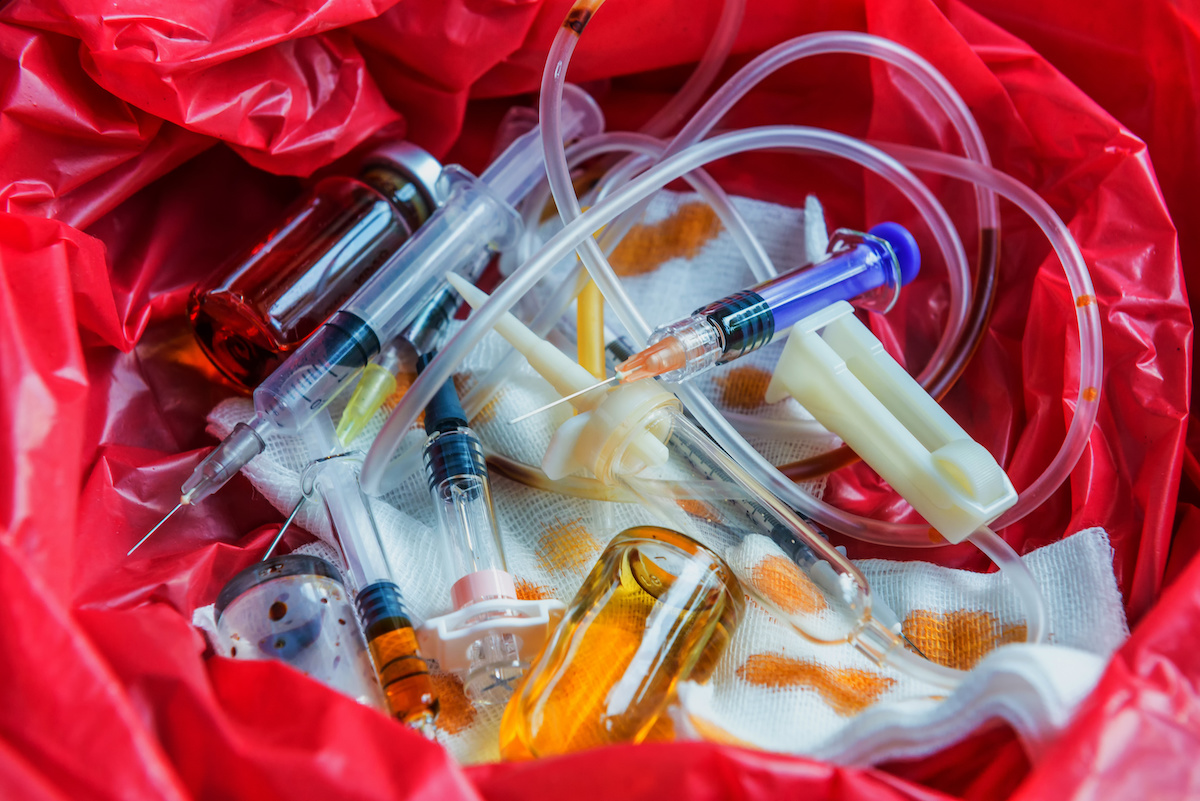
Correct Answer is B
Explanation
Choice A rationale: Plant pigments do not produce photon energy, but rather capture it from the sun. Photon energy is the energy carried by particles of light, called photons. Different types of electromagnetic radiation, such as visible light, have different amounts of photon energy depending on their wavelength¹.
Choice B rationale: Plant pigments absorb light energy and use it to initiate photosynthesis. Photosynthesis is the process by which plants convert light energy into chemical energy, stored in the bonds of sugar molecules. Plant pigments are specialized organic molecules, such as chlorophyll and carotenoids, that are found in the chloroplasts of plant cells. They absorb specific wavelengths of light and reflect others, giving plants their characteristic colors²³.
Choice C rationale: Plant pigments do not provide electrons, but rather transfer them to other molecules. Electrons are negatively charged subatomic particles that are involved in chemical reactions. In photosynthesis, plant pigments absorb light energy and use it to split water molecules, releasing electrons, protons, and oxygen. The electrons are then passed along an electron transport chain, generating a proton gradient that drives the synthesis of ATP, an energy molecule. The electrons are also used to reduce NADP+ to NADPH, an electron carrier⁴.
Choice D rationale: Plant pigments do not convert heat to electricity, but rather convert light to chemical energy. Heat and electricity are both forms of energy, but they are not directly involved in photosynthesis. Heat is the kinetic energy of molecules, while electricity is the flow of electrons or electric charge. Plant pigments absorb light energy and use it to drive the chemical reactions of photosynthesis, which produce sugar and oxygen as products⁵.
Choice E rationale: Plant pigments do not reduce NADP, but rather donate electrons to it. Reduction is a chemical reaction in which a molecule gains electrons, while oxidation is a chemical reaction in which a molecule loses electrons. NADP+ is an oxidized form of NADP, which stands for nicotinamide adenine dinucleotide phosphate. It is an electron carrier that accepts electrons from plant pigments in photosystem I, a complex of proteins and pigments in the thylakoid membrane of the chloroplast. The reduced form of NADP is NADPH, which carries electrons and hydrogen for the dark reaction of photosynthesis, which uses CO2 to produce glucose⁶.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
