Which of the following is correct regarding the pH scale?
A substance with a pH of 3 is two times more alkaline than a substance with a pH of 4.
A substance with a pH of 3 is two times more acidic than a substance with a pH of 4.
A substance with a pH of 3 is 10 times more acidic than a substance with a pH of 4.
A substance with a pH of 3 is 10 times more alkaline than a substance with a pH of 4.
The Correct Answer is C
The pH scale is a logarithmic scale that measures the acidity or alkalinity of a solution.
A solution with a pH of 7 is neutral, while a solution with a pH less than 7 is acidic and a solution with a pH greater than 7 is alkaline.
Because the pH scale is logarithmic, each whole number change in pH represents a tenfold change in acidity or alkalinity.
Therefore, a substance with a pH of 3 is 10 times more acidic than a substance with a pH of 4.
Choice A. A substance with a pH of 3 is two times more alkaline than a substance with a pH of 4 is not correct because it incorrectly states that the substance with a lower pH is more alkaline and also incorrectly states the magnitude of the difference in acidity or alkalinity.
Choice B. A substance with a pH of 3 is two times more acidic than a substance with a pH of 4 is not correct because it correctly states that the substance with a lower pH is more acidic but incorrectly states the magnitude of the difference in acidity.
Choice D. A substance with a pH of 3 is 10 times more alkaline than a substance with a pH of 4 is not correct because it incorrectly states that the substance with a lower pH is more alkaline.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is A
Explanation
The cell membrane is a thin, flexible barrier that surrounds all cells and separates the inside of the cell from the outside environment.
It is composed of a lipid bilayer and regulates the movement of substances into and out of the cell.
Choice B is incorrect because the Golgi apparatus is not present in prokaryotic cells.
The Golgi apparatus is an organelle found in eukaryotic cells that is involved in modifying, sorting, and packaging proteins and lipids for transport to other parts of the cell or to be secreted outside the cell.
Choice C is incorrect because chloroplasts are not present in prokaryotic cells.
Chloroplasts are organelles found in plant cells and some algae that are responsible for photosynthesis.
Choice D is incorrect because the endoplasmic reticulum is not present in prokaryotic cells.
The endoplasmic reticulum is an organelle found in eukaryotic cells that is involved in protein synthesis and lipid metabolism.
Correct Answer is D
Explanation
The pancreas secretes large amounts of sodium bicarbonate, which protects the duodenum by neutralizing the acid that comes from the stomach.
This compound helps neutralize stomach acid generated during the digestive process.
Choice A is incorrect because sodium bicarbonate is not a protease that digests carbohydrates.
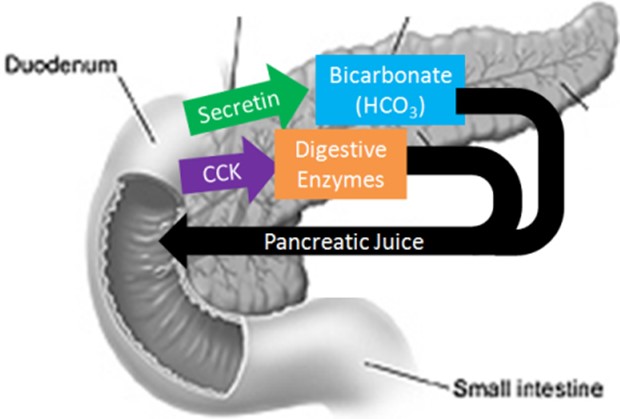
Proteases are enzymes that break down proteins, while sodium bicarbonate is a chemical compound that helps neutralize stomach acid.
Choice B is incorrect because sodium bicarbonate does not stimulate the pyloric sphincter.
The pyloric sphincter is a ring of smooth muscle that separates the stomach from the duodenum and regulates the passage of partially digested food (chyme) into the small intestine.
Choice C is incorrect because sodium bicarbonate does not inhibit peristalsis.
Peristalsis is a series of wave-like muscle contractions that move food through the digestive tract.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
