A decreased serum pH causes a condition known as:
Acidosis
Equal bicarbonate
Neutral carbonic acid
Alkalosis
The Correct Answer is A
Choice A reason: This is correct because acidosis is a condition where the serum pH is lower than the normal range of 7.35 to 7.45. Acidosis can be caused by an excess of acids or a loss of bases in the body, which can affect the function of various organs and systems.
Choice B reason: This is incorrect because equal bicarbonate is not a condition, but a term that describes the balance between bicarbonate (HCO3-) and carbonic acid (H2CO3) in the blood. Bicarbonate is a base that buffers the acids in the blood and maintains the pH. Equal bicarbonate means that the ratio of bicarbonate to carbonic acid is 20:1, which is the normal value.
Choice C reason: This is incorrect because neutral carbonic acid is not a condition, but a term that describes the pH of carbonic acid (H2CO3) in the blood. Carbonic acid is an acid that forms when carbon dioxide (CO2) dissolves in water. Neutral carbonic acid means that the pH of carbonic acid is 7.0, which is neither acidic nor basic.
Choice D reason: This is incorrect because alkalosis is a condition where the serum pH is higher than the normal range of 7.35 to 7.45. Alkalosis can be caused by a loss of acids or an excess of bases in the body, which can affect the function of various organs and systems.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is D
Explanation
Choice A reason: Stroke is not a cause of hyponatremia, but rather a possible complication of it. Hyponatremia is a condition where the sodium level in the blood is too low, which can affect the brain function and cause symptoms such as confusion, seizures, or coma. Stroke is a condition where the blood supply to a part of the brain is interrupted, which can cause brain damage and neurological deficits.
Choice B reason: Dehydration is not a cause of hyponatremia, but rather a cause of hypernatremia. Dehydration is a condition where the body loses more fluids than it takes in, which can affect the blood volume and the electrolyte balance. Dehydration can cause hypernatremia, which is a condition where the sodium level in the blood is too high, which can also affect the brain function and cause symptoms such as thirst, dry mouth, or lethargy.
Choice C reason: Increased secretion of aldosterone is not a cause of hyponatremia, but rather a cause of hypokalemia. Aldosterone is a hormone that regulates the sodium and potassium levels in the body by increasing the reabsorption of sodium and the excretion of potassium in the kidneys. Increased secretion of aldosterone can cause hypokalemia, which is a condition where the potassium level in the blood is too low, which can affect the muscle and nerve function and cause symptoms such as weakness, cramps, or arrhythmias.
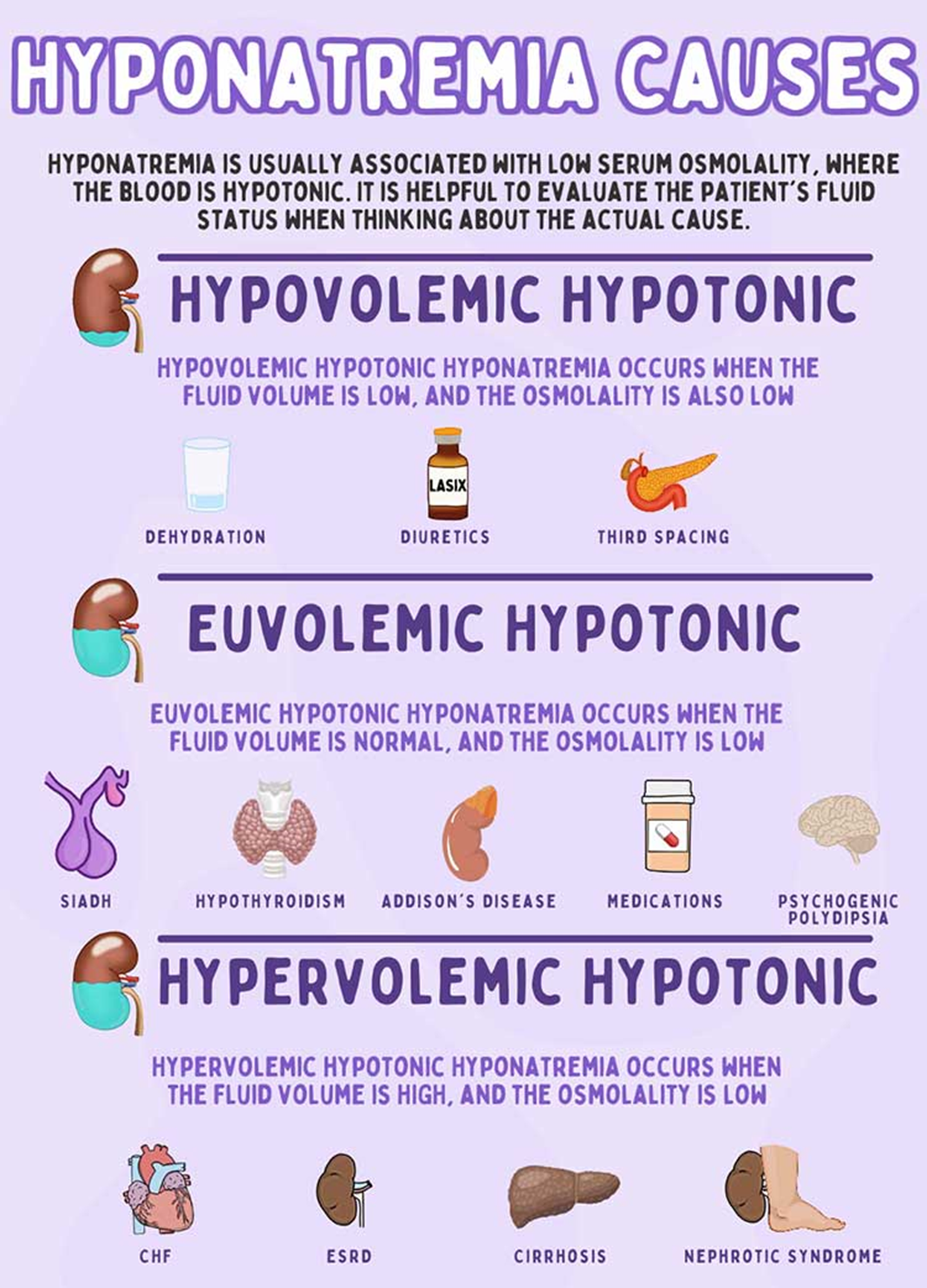
Choice D reason: Congestive heart failure (CHF) is a cause of hyponatremia, as it is a condition where the heart is unable to pump enough blood to meet the body's needs. This can lead to fluid retention and edema, which can dilute the sodium level in the blood and cause hyponatremia. CHF can also stimulate the release of antidiuretic hormone (ADH), which increases the reabsorption of water in the kidneys and further lowers the sodium level in the blood.
Correct Answer is A
Explanation
Choice A reason: This is correct because respiratory acidosis is a condition where the blood pH is lower than the normal range of 7.35 to 7.45, due to impaired gas exchange or hypoventilation, which causes carbon dioxide to accumulate in the blood. COPD is a chronic lung disease that obstructs the airways and reduces the oxygen intake and carbon dioxide output. This leads to respiratory acidosis in the patient.
Choice B reason: This is incorrect because respiratory alkalosis is a condition where the blood pH is higher than the normal range of 7.35 to 7.45, due to hyperventilation, which lowers the carbon dioxide in the blood. COPD does not cause hyperventilation, but rather hypoventilation.
Choice C reason: This is incorrect because metabolic alkalosis is a condition where the blood pH is higher than the normal range of 7.35 to 7.45, due to a loss of acids or an excess of bases in the body. COPD does not affect the metabolic system directly, but rather the respiratory system.
Choice D reason: This is incorrect because metabolic acidosis is a condition where the blood pH is lower than the normal range of 7.35 to 7.45, due to an excess of acids or a loss of bases in the body. COPD does not affect the metabolic system directly, but rather the respiratory system.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
