The pleura is a connective tissue sheath that covers which of the following organs?
Liver.
Heart.
Spleen.
Lung.
The Correct Answer is D
The pleura is a double-layered serous membrane that covers each lung and lines the thoracic cage.
The pleura is a vital part of the respiratory tract.
Its role is to cushion the lung and reduce any friction that may develop between the lung, rib cage, and chest cavity.
Each pleura (there are two) consists of a two-layered membrane that covers each lung.
The layers are separated by a small amount of viscous (thick) lubricant known as pleural fluid.
The pleura is comprised of two distinct layers: the visceral pleura and the parietal pleura.
The visceral pleura is the thin, slippery membrane that covers the surface of the lungs and dips into the areas separating the different lobes of the lungs (called the hilum).
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is D
Explanation
Proteins are made up of amino acids which are organic molecules that contain both an amine functional group (–NH2) and a carboxylic acid functional group (– COOH)1.
Choice A, Lipids, is not the correct answer because lipids are a group of naturally occurring molecules that include fats, waxes, sterols, fat-soluble vitamins, monoglycerides, diglycerides, triglycerides, phospholipids, and others.
They do not contain both an amine and carboxyl group.
Choice B, Chitin, is not the correct answer because chitin is a long-chain polymer of N-acetylglucosamine, a derivative of glucose.
It does not contain both an amine and carboxyl group.
Choice C, Cellulose, is not the correct answer because cellulose is an organic compound with the formula (C6H10O5)n, a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked D-glucose units.
It does not contain both an amine and carboxyl group.
Correct Answer is D
Explanation
Genes are segments of DNA that encode the information for making proteins.
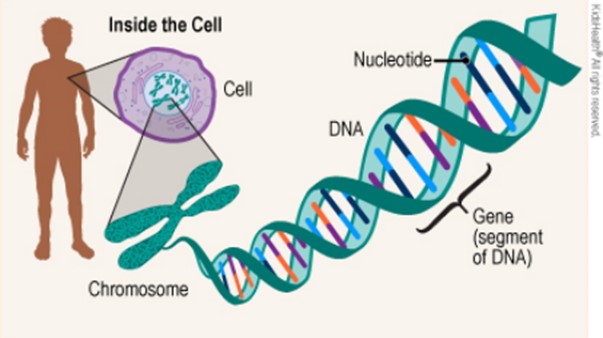
The sequence of nucleotides (As, Ts, Cs, and Gs) in a gene determines the amino acid sequence of the protein. DNA sequencing is the process of determining the sequence of nucleotides in a piece of DNA.
Choice A is incorrect because enzymes are proteins that catalyze chemical reactions, not DNA sequences. Choice B is incorrect because blood types are determined by the presence or absence of certain antigens on the surface of red blood cells, not by DNA sequences. Choice C is incorrect because hormones are chemical messengers that regulate various body functions, not DNA sequences.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
