A myocardial infarction affects which of the following blood vessels of the heart?
Coronary
Aorta
Pulmonary
Vena cava.
The Correct Answer is A
A myocardial infarction, commonly known as a heart attack, occurs when blood flow decreases or stops in the coronary artery of the heart, causing damage to the heart muscle.
Choice B is incorrect because the aorta is not a blood vessel of the heart.
The aorta is the main artery that carries oxygenated blood from the heart to the rest of the body.
Choice C is incorrect because the pulmonary blood vessels are not affected by a myocardial infarction.
The pulmonary blood vessels carry deoxygenated blood from the heart to the lungs.
Choice D is incorrect because the vena cava is not a blood vessel of the heart.
The vena cava is a large vein that carries deoxygenated blood from the body to the heart.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is D
Explanation
Nitrogen gas (N2) is an extremely stable molecule because it consists of two nitrogen atoms bonded together by a triple covalent bond.
A covalent bond is a type of chemical bond where atoms share electrons to form a molecule.
In a triple covalent bond, three pairs of electrons are shared between the two atoms, resulting in a very strong bond that makes the molecule extremely stable.
Choice A. Ionic bonds is not correct because ionic bonds involve the transfer of electrons from one atom to another to form ions, which are then attracted to each other due to their opposite charges.
Nitrogen gas does not contain ions and is not held together by ionic bonds.
Choice B. Hydrogen bonds is not correct because hydrogen bonds are weak electrostatic attractions between molecules that contain hydrogen atoms bonded to highly electronegative atoms such as oxygen or nitrogen.
Nitrogen gas does not contain hydrogen atoms and is not held together by hydrogen bonds.
Choice C. Resonance bonds is not correct because resonance refers to the delocalization of electrons in a molecule where multiple Lewis structures can be drawn to represent the molecule.
Nitrogen gas has a single Lewis structure and does not exhibit resonance.
Correct Answer is B
Explanation
The atomic number of an atom is equal to the number of protons in its nucleus. In this case, the atom has 12 protons, so its atomic number is.
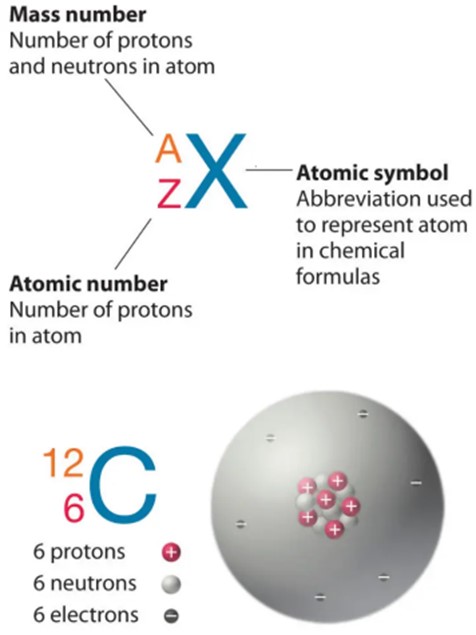
Choice A, 24, is not the correct answer because it represents the sum of the number of protons and neutrons in the atom’s nucleus, which is known as the mass number.
Choice C, 1, is not the correct answer because it does not represent the number of protons in the atom’s nucleus.
Choice D, 144, is not the correct answer because it represents the square of the mass number and does not represent any property of the atom.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
