A patient with a gastric ulcer is ordered sucralfate (Carafate). How does this medication works?
calm the patient to reduce acid production
block the H2 receptors
neutralize the gastric acids
coat the gastric lining
The Correct Answer is D
A. Calm the patient to reduce acid production.
This description is not accurate for sucralfate. Calming the patient to reduce acid production is typically associated with medications like proton pump inhibitors (PPIs) or H2 receptor blockers.
B. Block the H2 receptors.
Blocking H2 receptors is the mechanism of action for H2 receptor blockers, such as ranitidine. It is not the mechanism of action for sucralfate.
C. Neutralize the gastric acids.
Neutralizing gastric acids is the mechanism of action for antacids, such as aluminum hydroxide or calcium carbonate. Sucralfate works differently; it forms a protective coating on the gastric lining rather than directly neutralizing acids.
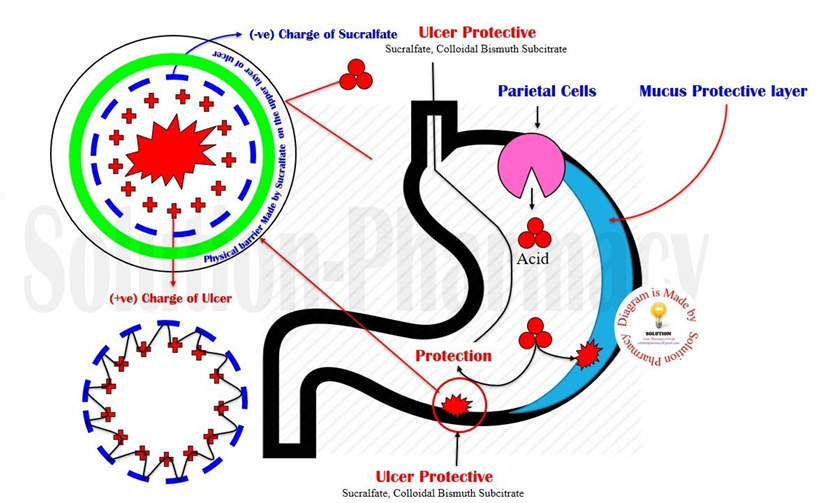
D. Coat the gastric lining.
This is the correct mechanism of action for sucralfate. It forms a protective coating on the gastric lining, adhering to the ulcer site and providing a barrier against gastric acid.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is A
Explanation
A. Misoprostol (Cytotec)
The patient complaining of gastric distress from NSAIDs (Nonsteroidal Anti-Inflammatory Drugs) like aspirin or indomethacin may benefit from the administration of misoprostol (Cytotec). Misoprostol is a prostaglandin analog that helps protect the stomach lining and reduce the risk of NSAID-induced gastric ulcers.
B. Lansoprazole (Prevacid)
Lansoprazole is a proton pump inhibitor (PPI) that reduces stomach acid production. While PPIs can be used for certain acid-related conditions, they do not directly protect against NSAID-induced gastric distress.
C. Magaldrate (Riopan)
Magaldrate is an antacid that neutralizes stomach acid. It may provide relief from symptoms of indigestion but does not specifically address the gastric distress caused by NSAIDs.
D. Magnesium trisilicate (Gaviscon)
Magnesium trisilicate is an antacid that helps neutralize stomach acid. Like magaldrate, it may alleviate symptoms of indigestion but does not target the underlying issue of NSAID-induced gastric distress.
Correct Answer is A
Explanation
A. The importance of consuming adequate amounts of water
When instructing a patient who will take psyllium (Metamucil) to treat constipation, it is crucial to emphasize the importance of consuming adequate amounts of water. Psyllium is a bulk-forming laxative that works by absorbing water in the intestines, forming a gel-like mass that helps soften stools and promote bowel movements. Without sufficient water intake, psyllium may cause an obstruction or worsen constipation.
B. The need to monitor for systemic side effects.
Psyllium is generally well-tolerated and is not associated with significant systemic side effects. The primary consideration is ensuring proper hydration.
C. The onset of action of 30-60 minutes after administration.
Psyllium is not a fast-acting laxative. It usually takes a longer time to produce its effects, and patients should not expect an immediate response within 30-60 minutes.
D. The need to use the dry form of psyllium to prevent cramping.
Using the dry form of psyllium is not recommended, as it may increase the risk of choking or swallowing difficulties. It is typically recommended to mix psyllium with a sufficient amount of water or other liquids to prevent cramping and ensure proper absorption.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
